Translate this page into:
Mycobacterium chimaera: A novel pathogen with potential risk to cardiac surgical patients
2 Head of Clinical Laboratory, Belle Vue Clinic, Hospital Infection Control and Quality Assurance, Kolkata, West Bengal, India
3 Department of Microbiology, Christian Medical College, Vellore, Tamil Nadu, India
Corresponding Author:
Ruaridh Buchanan
Division of Infection, Royal London Hospital, Barts Health NHS Trust, 80 Newark Street, London, E1 2ES
United Kingdom
ruaridhbuchanan@doctors.org.uk
| How to cite this article: Buchanan R, Agarwal A, Mathai E, Cherian BP. Mycobacterium chimaera: A novel pathogen with potential risk to cardiac surgical patients. Natl Med J India 2020;33:284-287 |
Abstract
Mycobacterium chimaera was first described in 2004, coming to prominence in 2011 with reports from across the globe of invasive infections following cardiac surgery. This outbreak was linked to a specific type of heater cooler machine used for cardiac surgery by whole-genome sequencing. We briefly outline what is currently known about this pathogen, highlighting the importance of clinical vigilance and the diagnostic options for the clinician.Introduction
‘One-third of heart devices contaminated with bacteria’ read the headlines in summer 2017.[1] This was how the mainstream media reported a presentation to the 44th Annual Conference of the Association for Professionals in Infection Control and Epidemiology in the USA, which described the discovery of non-tuberculous mycobacteria in heater cooler units (HCUs), a component of cardiac bypass machinery, in a number of countries. The United States Centers for Disease Control and Prevention (CDC)[2] and Food and Drug Administration (FDA), along with their counterparts in Europe and Australia, have all issued safety alerts regarding the risks of cardiopulmonary bypass—specifically, that of infection with Mycobacterium chimaera (M. chimaera).
‘Health tourism’ is a major business in India—it is estimated that 1 million people visited India for healthcare in 2015, contributing US$ 3 billion to the economy.[3] India is a popular destination for cardiac surgery due to its skilled surgical teams and high-quality clinical facilities; there are currently around 700 cardiac surgeons performing upwards of 150 000 procedures every year.[4] Despite the large numbers of procedures, to date, there are no published reports of M. chimaera infections in India—concerns have been raised about the possibility of underreporting of the infection in low- and middle-income countries, either due to lack of awareness or lack of access to the necessary diagnostic investigations.[5]
Compared with healthcare systems in other major economies, Indian providers have different challenges to face. Infection prevention and control remains an emerging field, complicated by the complexities of the healthcare infrastructure and budget allocation. We aim to raise the profile of this infection so that physicians, surgeons, microbiologists and infection specialists may consider this in the differential diagnosis in patients at risk.
The Pathogen
M. chimaera is a species which forms part of the Mycobacterium avium/intracellulare complex (MAC). First identified as its own species in 2004,[6] M. chimaera is so named due to its genome comprising elements from three different species within the complex. It is phenotypically indistinguishable from other members of the complex, making its identification a challenge for laboratories without access to advanced diagnostic techniques—discussed in detail below.
A Global Outbreak
The first case of M. chimaera endocarditis was reported in Zürich in 2013 among a series of cases dating back to 2011.[7] A further six cases were then reported from Switzerland in 2015,[8] followed by reports of at least 15 cases in Germany[9],[10] as well as reports from the USA,[11] the UK,[12] Australia and New Zealand[13] among other countries. Epidemiological investigation has strongly implicated contaminated HCUs as the source of the infection.[9] Since the outbreak gained international recognition, publication of new cases has slowed, but active case finding continues—public health agencies in the UK and many other countries are engaged in programmes of active surveillance, as well as taking control measures to limit the risk of further infections. Estimates of risk vary, with Public Health England (PHE) estimating the risk of infection at approximately 1:5000 for valve replacements and 1:10 000 for other cardiac surgeries[12] with risk directly proportional to time spent on bypass.[11]
Clinical Presentation
Patients have presented with features from as early as 3 months to as late as 5 years following cardiac surgery.[12] Clinical cases can be grouped into four main syndromes:
- Wound infection: This most often affects the sternotomy wound, causing wound breakdown, sternal osteomyelitis and sometimes hyperostosis. However, we also have experience of infection at the site of saphenous vein extraction (case not published), presenting with chronic and recurrent cellulitis and associated with abscess formation and local lymphadenopathy.
- Pulmonary infection: This presentation is identical to that of other MAC infections. Patients may suffer from chronic cough and breathlessness, with or without associated constitutional features such as fatigue, fever, night sweats and weight loss. Chest imaging may reveal nodules and hilar lymphadenopathy—several patients have been erroneously diagnosed with sarcoidosis or pulmonary tuberculosis before the diagnosis of M. chimaera infection was made.[10]
- Disseminated infection: In these patients, constitutional symptoms predominate, with multifocal lymphadenopathy and hepatosplenomegaly found on examination; bone marrow infiltration can result in pancytopenia. There are also reports of immune thrombocytopenia[14] and uveitis[15] as manifestations of the disease, as well as cerebral vasculitis,[10] hepatitis and nephritis.[8] Lack of specific clinical syndromes unique to M. chimaera infections poses a major challenge for diagnosis.
- Infective endocarditis: The presentation is that of endocarditis of any cause but is more likely to affect prosthetic valves and can take a fulminant course with several fatal cases reported.[10] It is culture-negative using routine culture media; mycobacterial cultures are not routinely pursued in this clinical setting; thus, the diagnosis may be missed.
Diagnosis
Investigations required to make the diagnosis depend on the clinical presentation of the individual patient. In the case of wound infection, obtaining tissue for mycobacterial culture and histopathology are the key to confirming the diagnosis. In one series, culture from sternal wound tissue samples was positive for M. chimaera in 100% of cases;[16] histopathological appearances of non-necrotizing granulomata have been found in approximately 79% of biopsy samples,[16] whereas acid-fast bacilli are rarely seen.
For pulmonary disease, sputum samples or targeted deeper sampling with lavage of respiratory secretions can yield the diagnosis, as in all pulmonary MAC infection.[17] In patients with non-specific constitutional symptoms, cross-sectional imaging may identify enlarged lymph nodes for biopsy.
Given that patients were exposed to M. chimaera during cardiac surgery and that endocarditis is one of the most serious manifestations of infection, echocardiography is a priority investigation. Transthoracic echocardiography has been shown to have a sensitivity of 67%, lower than that of transoesophageal echocardiography;[16] the latter approach should be favoured in cases where infective endocarditis is suspected.
In all suspected cases, mycobacterial blood cultures should be done, with their yield higher in patients suffering from infective endocarditis than any other syndrome, reaching a sensitivity of 64% in untreated patients, though this is halved following the initiation of appropriate therapy.[16] Bone marrow culture has been shown to have a sensitivity of up to 80%, though this may be an overestimate given the severity of the disseminated cases reported in the literature.[16]
Laboratory Identification
M. chimaera is a slow-growing, non-chromogenic mycobac-terium within the MAC, identified as a unique species in 2004.[6] It can be grown using standard mycobacterial culture methods; however, M. chimaera cannot be differentiated from the other members of the complex by phenotypic methods. It is easily identified by polymerase chain reaction,[18] and some, but not all, commercial mycobacterial identification line probe assays can also identify M. chimaera to the species level. Other assays, including current MALDI-TOF MS databases, would initially mis-identify it as M. intracellulare,[18] though updates now allow for more reliable differentiation.[19] In a patient with suspected M. chimaera infection, all mycobacterial isolates should ideally be referred to a laboratory capable of identifying non-tuberculous mycobacteria to the species level, including M. chimaera. Clinicians must be aware of the limitations of their local laboratory and of the possibility of mis-identification as other non-tuberculous mycobacteria. This is especially true for India, where access to quality assured mycobacterial cultures, including mycobacterial blood cultures, may not be universal. Seeking help from specialist laboratories may be an interim solution while developing in-house laboratory capacity.
Clinical Management
There are as yet insufficient data to ascertain the best drug regimens and length of treatment for M. chimaera infections. Most centres have elected to follow the current American[20] or British[17] consensus guidelines for treating MAC infections. The combination of rifampicin, ethambutol and a macrolide is most commonly used, with the addition of intravenous amikacin in more severe infections. Alternative agents include fluoro-quinolones and clofazimine. A minimum of 12 months of therapy after culture conversion has been suggested, but data are lacking. It is worth noting that patients at risk of M. chimaera infection pose numerous challenges for pharmacotherapy. They are likely to be older patients, thus at a greater risk of side-effects such as rifampicin-induced hepatitis or loss of vision due to ethambutol. Almost all will have significant cardiovascular comorbid conditions, increasing the risk of renal impairment with amikacin and rhythm disturbance with macrolides and fluoroquinolones. They are also more likely to be on multiple medications, with an increased risk of drug interactions, particularly with the administration of potent liver enzyme modifiers such as rifampicin and macrolides. Therefore, an individualized approach to therapy is likely to yield better results than strict adherence to broad guidelines.
Definitive management also depends on the site of infection and suitability of surgical management based on the limited case series reported so far. Cases of infective endocarditis have required prompt surgical intervention to achieve good outcomes.
Controlling the Environmental Reservoir
Molecular epidemiological studies confirmed that the M. chimaera isolates from patients affected by the outbreak were identical to those obtained from Stöckert 3T HCUs manufactured by Sorin Group Deutschland GmbH (now LivaNova PLC). Identical isolates were subsequently found in the water at the company’s manufacturing and maintenance plant. It was, thus, concluded that there was a high likelihood that new HCUs being shipped to hospitals were already contaminated with M. chimaera.[9]
HCUs are used during cardiopulmonary bypass, when the patient’s heart is prevented from beating and the circulation maintained by external mechanical pumping of blood. Blood and cardioplegia fluid are transported through lengths of tubing which would normally result in both rapidly assuming room temperature. This is prevented by HCUs, which contain individual water baths maintained at cold and warm temperatures. The bypass circuit tubing passes through these baths, keeping the blood and cardioplegia fluid at their required temperatures. There is no direct contact between the water in the HCU and the fluid within the tubing; however, the HCU regulates its temperature using fans which can act to aerosolize water from the unit. It has been demonstrated that the vapour generated by HCUs can rapidly penetrate the air curtain surrounding the patient in ultraclean-ventilated operating theatres, exposing the patient to aerosolized M. chimaera [Figure - 1].[21]
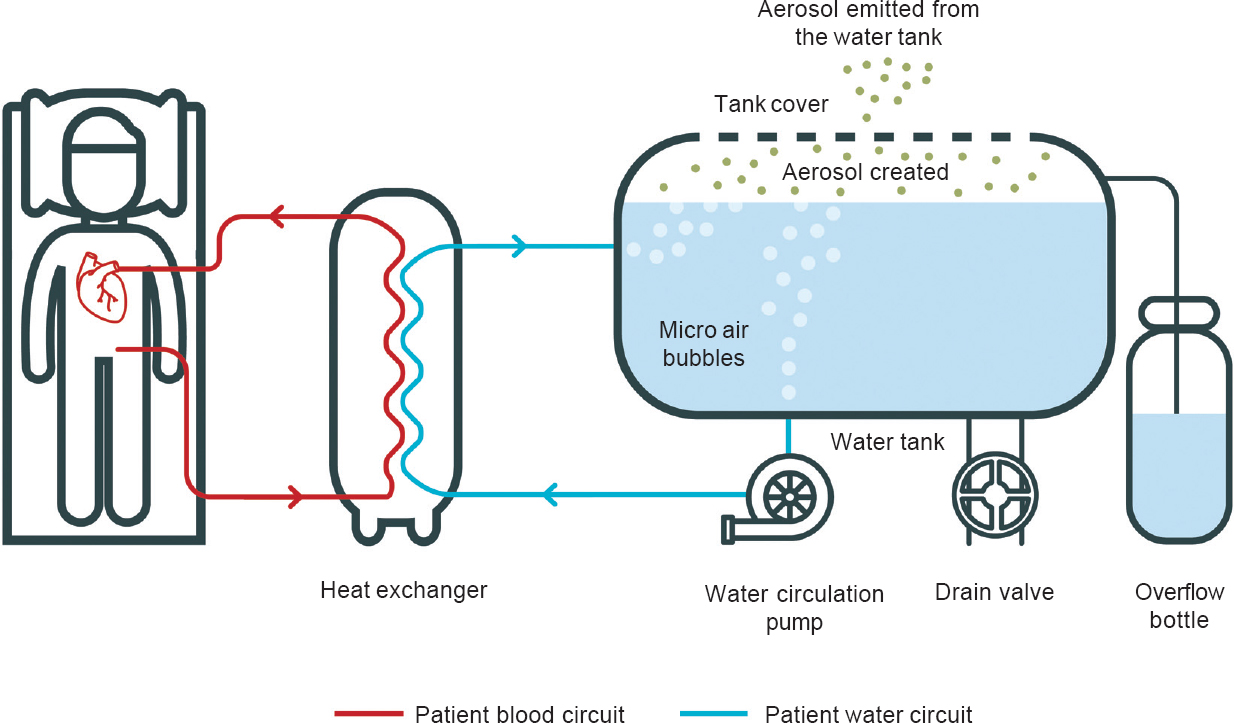 |
| Figure 1: Heater cooler unit schematic showing generation of aerosol (with thanks to Iain Morrison, IDEAM, Edinburgh) |
Globally, there are five market leaders supplying HCUs including Sorin/LivaNova. There is neither publicly available information on the numbers supplied nor the distribution destinations of each model; however, we are aware that at least two Indian centres serving a large metropolitan area use the affected Stöckert 3T model.
Prevention
Ensuring that HCUs are free of M. chimaera and remain so is the key to controlling the outbreak. Manufacturers provide instructions on cleaning and decontamination—these guidelines have been updated since the beginning of the outbreak. Biofilm formation within the circuit is a major issue and replacing these pipes has been an effective strategy in reducing the bioburden within the system. There are reports that daily application of 3% hydrogen peroxide, in combination with biweekly full decontamination with peracetic acid, is effective in reducing viable M. chimaera;[22] however, this aggressive approach has been shown to cause degradation of elements of the HCU, requiring more frequent repair.[23] There is debate about the utility of regular surveillance cultures from HCUs given the time delay between sampling and culture positivity; however, it remains the standard of practice within some centres, including our own, and is recommended by PHE, UK.[24] A practical minimum approach would seem to be thorough cleaning and disinfection fully compliant with the manufacturer’s instructions, supervised by a named responsible individual, until such time as more definitive evidence-based guidelines become available. This may involve a considerable change in practice—a poll of three major cardiac surgery units in Indian metropolitan areas suggests that current cleaning practices do not meet the recommended standard. There is currently no consensus among experts on the utility of environmental sampling including water testing; individual centres will have to carry out a risk assessment and consider performing regular sampling to monitor microbial contamination of HCU water.
An alternative preventive measure is to place physical barriers between the HCU and the operative field. Exhaust air from Sorin HCUs is ejected from the rear of the device; so, ensuring that this side faces away from the patient can in theory reduce the risk of contamination. Placing the unit inside a specially constructed case with exhaust tubing stretching outside the operating theatre has also been tried and is thought to reduce this risk to the patient, though there are as yet no data to support this. This approach is taken to its logical extreme by removing the HCU from the operating theatre entirely, using longer tubing to connect it to the rest of the perfusion circuit and forming an aperture in the theatre wall to allow passage of this tubing. These strategies have been adopted in some centres but are not without their own issues as the HCUs were neither designed to be encased nor to pump fluid over long distances; there is also the additional trip hazard associated with increased tubing and potential for disturbance of operating theatre positive pressure ventilation. Current PHE guidance recommends placing the HCU outside of the operating theatre only if safe and practical to do so.[25] Any attempt to change the direction of exhaust air or aerosols from HCUs also needs to take into account the pressure differentials and air changes within the operating theatre, be it an ultraclean-ventilated theatre or a more conventionally ventilated space—liaison with clinical engineering colleagues is recommended before making any major changes.
Conclusion
Cases of M. chimaera infection linked to contaminated HCUs have been reported around the world; considering the lengthy incubation period, new cases are likely to be identified over the next several years. Given the high numbers of cardiac surgeries performed in India each year, it is likely that there will be cases of M. chimaera infection presenting to Indian physicians. An awareness of the risk of this infection and its myriad presentations will allow physicians to identify potential cases and undertake appropriate investigations and treatment. An understanding of the capabilities of the local microbiology laboratory with regard to mycobacterial identification is necessary, and close communication with microbiology and histopathology colleagues is needed to ensure that correct investigations are undertaken. Cardiac surgery centres using the affected HCUs should take steps to ensure that the units are being tested, decontaminated and serviced in accordance with the most up-to-date guidance, with consideration given to undertaking active case finding among their cohort of patients.
Conflicts of interest. None declared
| 1. | One-third of heart surgery devices contaminated with bacteria. Hindustan Times; 2017. Available at www.hindustantimes.com/fitness/one-third-of-heart-surgeryd evices-contaminated-with-bacteria-study/story-Ep3JUfDvUMjh0 NXW6rz16L.html (accessed on 18 Jan 2018). [Google Scholar] |
| 2. | Perkins KM, Lawsin A, Hasan NA, Strong M, Halpin AL, Rodger RR, et al. Notes from the field: Mycobacterium chimaera contamination of heater-cooler devices used in cardiac surgery—United States. MMWR Morb Mortal Wkly Rep 2016;65: 1117–18. [Google Scholar] |
| 3. | Bhabani S. Number of medical tourists coming from India has more than doubled since 2013. Mail Online India; 2016. Available at www.dailymail.co.uk/indiahome/indianews/article-3726175/Number-medical-tourists-coming-India-doubles-two-years.htm (accessed on 18 Jan 2018). [Google Scholar] |
| 4. | Hosain N, Amin F, Rehman S, Koirala B. Know thy neighbors: The status of cardiac surgery in the South Asian countries around India. Indian Heart J 2017;69:790–6. [Google Scholar] |
| 5. | Ortiz-Martínez Y. Mycobacterium chimaera: An under-diagnosed pathogen in developing countries? J Hosp Infect 2017;97:125–6. [Google Scholar] |
| 6. | Tortoli E, Rindi L, Garcia MJ, Chiaradonna P, Dei R, Garzelli C, et al. Proposal to elevate the genetic variant MAC-A, included in the Mycobacterium avium complex, to species rank as Mycobacterium chimaera sp. nov. Int J Syst Evol Microbiol 2004;54:1277–85. [Google Scholar] |
| 7. | Achermann Y, Rössle M, Hoffmann M, Deggim V, Kuster S, Zimmermann DR, et al. Prosthetic valve endocarditis and bloodstream infection due to Mycobacterium chimaera. J Clin Microbiol 2013;51:1769–73. [Google Scholar] |
| 8. | Sax H, Bloemberg G, Hasse B, Sommerstein R, Kohler P, Achermann Y, et al. Prolonged outbreak of Mycobacterium chimaera infection after open-chest heart surgery. Clin Infect Dis 2015;61:67–75. [Google Scholar] |
| 9. | Haller S, Höller C, Jacobshagen A, Hamouda O, Abu Sin M, Monnet DL, et al. Contamination during production of heater-cooler units by Mycobacterium chimaera potential cause for invasive cardiovascular infections: Results of an outbreak investigation in Germany, April 2015 to February 2016. Euro Surveill 2016;21:pii=30215. [Google Scholar] |
| 10. | Kohler P, Kuster SP, Bloemberg G, Schulthess B, Frank M, Tanner FC, et al. Healthcare-associated prosthetic heart valve, aortic vascular graft, and disseminated Mycobacterium chimaera infections subsequent to open heart surgery. Eur Heart J 2015;36:2745-53. [Google Scholar] |
| 11. | Lyman MM, Grigg C, Kinsey CB, Keckler MS, Moulton-Meissner H, Cooper E, et al. Invasive nontuberculous mycobacterial infections among cardiothoracic surgical patients exposed to heater-cooler devices. Emerg Infect Dis 2017;23:796–805. [Google Scholar] |
| 12. | Chand M, Lamagni T, Kranzer K, Hedge J, Moore G, Parks S, et al. Insidious risk of severe Mycobacterium chimaera infection in cardiac surgery patients. Clin Infect Dis 2017;64:335–42. [Google Scholar] |
| 13. | Karim F, Clahsen-van Groningen M, van Laar JA. AA amyloidosis and IgG4-related disease. N Engl J Med 2017;376:599–600. [Google Scholar] |
| 14. | Sacco KA, Burton MC. Persistent immune thrombocytopenia heralds the diagnosis of Mycobacterium chimaera prosthetic valve endocarditis. ID Cases 2017;7:1–3. [Google Scholar] |
| 15. | Zweifel SA, Mihic-Probst D, Curcio CA, Barthelmes D, Thielken A, Keller PM, et al. Clinical and histopathologic ocular findings in disseminated Mycobacterium chimaera infection after cardiothoracic surgery. Ophthalmology 2017;124:178–88. [Google Scholar] |
| 16. | Scriven JE, Scobie A, Verlander NQ, Houston A, Collyns T, Cajic V, et al. Mycobacterium chimaera infection following cardiac surgery in the United Kingdom: Clinical features and outcome of the first 30 cases. Clin Microbiol Infect 2018;24:1164–70. [Google Scholar] |
| 17. | Haworth CS, Banks J, Capstick T, Fisher AJ, Gorsuch T, Laurenson IF, et al. British Thoracic Society guidelines for the management of non-tuberculous mycobacterial pulmonary disease (NTM-PD). Thorax 2017;72:ii1–64. [Google Scholar] |
| 18. | Lecorche E, Haenn S, Mougari F, Kumanski S, Veziris N, Benmansour H, et al. Comparison of methods available for identification of Mycobacterium chimaera. Clin Microbiol Infect 2018;24:409–13. [Google Scholar] |
| 19. | Epperson LE, Timke M, Hasan NA, Godo P, Durbin D, Helstrom NK, et al. Evaluation of a novel MALDI biotyper algorithm to distinguish Mycobacterium intracellulare from Mycobacterium chimaera. Front Microbiol 2018;9:3140. [Google Scholar] |
| 20. | Griffith DE, Aksamit T, Brown-Elliott BA, Catanzaro A, Daley C, Gordin F, et al. An official ATS/IDSA statement: Diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am J Respir Crit Care Med 2007;175:367–416. [Google Scholar] |
| 21. | Walker JT, Lamagni T, Chand M. Evidence that Mycobacterium chimaera aerosols penetrate laminar airflow and result in infections at the surgical field. Lancet Infect Dis 2017;17:1019. [Google Scholar] |
| 22. | Garvey MI, Ashford R, Bradley CW, Bradley CR, Martin TA, Walker J, et al. Decontamination of heater-cooler units associated with contamination by atypical mycobacteria. J Hosp Infect 2016;93:229–34. [Google Scholar] |
| 23. | Garvey MI, Bradley CW, Walker J. A year in the life of a contaminated heater-cooler unit with Mycobacterium chimaera? Infect Control Hosp Epidemiol 2017;38:705–11. [Google Scholar] |
| 24. | Kranzer K, Ahmed N, Hoffman P, Pink F. Protocol for environmental sampling, processing and culturing of water and air samples for the isolation of slowgrowing mycobacteria standard operating procedure. PHE Publications Gateway Number: 2016150 2 Protocol for Environmental Sampling; 2016. Available at www.gov.uk/ phe%0Awww.facebook.com/PublicHealthEngland (accessed on 18 Jan 2018). [Google Scholar] |
| 25. | Public Health England. Infections associated with heater cooler units used in cardiopulmonary bypass and ECMO: Information for healthcare providers in England about Public Health England; 2015. Available at www.gov.uk/government/uploa ds/system/uploads/at tachment_d ata/file/4 74218/ Infections_associated_with_heater_cooler_units_version_1.pdf . (accessed on 18 Jan 2018). [Google Scholar] |
Fulltext Views
2,898
PDF downloads
1,841




