Translate this page into:
Pharmacotherapy of patients with stable bronchial asthma
Corresponding Author:
Ashutosh Nath Aggarwal
Department of Pulmonary Medicine, Postgraduate Institute of Medical Education and Research, Chandigarh 160012
India
aggarwal.ashutosh@outlook.com
| How to cite this article: Aggarwal AN. Pharmacotherapy of patients with stable bronchial asthma. Natl Med J India 2016;29:330-337 |
Abstract
Several reliever and controller medications are available for managing patients with stable asthma. Inhaled corticosteroids (ICS) are the mainstay of treatment, and can be combined with long-acting beta-agonists (LABA) for patients with moderate or severe disease. An ICS-LABA combination is best administered as a single inhaler used for both maintenance and emergency use. A stepwise approach to therapy has recently been proposed as part of Indian guidelines for management of asthma. Clinicians should advise proper use of inhaled medications, and step up or step down treatment based on adequacy of asthma control.Introduction
Treatment of bronchial asthma is aimed at providing symptomatic relief to patients, and preventing disease progression by limiting airway inflammation. A properly tailored prescription leads to adequate asthma control and enables the patient to continue to carry out all day-to-day, work-related and recreational activities normally. It is equally essential to design a prescription that safeguards patients from any treatment-related adverse events. Although pharmacotherapy is currently the mainstay for achieving these goals, other areas such as patient education and avoidance of environmental triggers are also important.
Several international guidelines have outlined management principles for patients with stable bronchial asthma. [1] ,[2] ,[3] ,[4] ,[5] However, management of asthma continues to be suboptimal in India. This is attributed both to patients′ reluctance or inability to adhere to standard therapy, as well as physicians′ ignorance about recent evidence-based modifications in principles of treatment. To standardize diagnostic and management protocols for Indian patients, the Indian Chest Society and National College of Chest Physicians (India) supported a workshop of experts in 2014 to frame consensus guidelines. These guidelines were published in 2015, and are based on quality evidence gathered across the world and in India. [6] This review is based on the discussions and recommendations that resulted in the formulation of these management guidelines; it focuses on pharmacotherapeutic options currently available in India.
Basic Definitions
Pharmacological agents useful in managing asthma can be categorized as either ′controllers′ or ′relievers′. Controller medications are anti-inflammatory drugs (inhaled corticosteroids [ICS], leukotriene antagonists, mast cell stabilizers, etc.) or long-acting bronchodilators, and are responsible for control of airway inflammation. They need to be administered on a regular basis, even if the patient remains asymptomatic. In contrast, reliever medications (also termed rescue medications) relieve acute symptoms but do not control airway inflammation. They should therefore be taken only on an as-needed basis (not regularly) for immediate relief during episodes of symptomatic worsening. Bronchodilators with rapid onset of action are the commonest relievers.
Reliever Medications
Several inhaled agents can be used as reliever medicines on an as-needed basis, either alone or in combination, for managing patients with asthma. These include (i) short-acting beta-agonists (SABA) such as salbutamol, levosalbutamol and terbutaline; (ii) long-acting beta-agonists (LABA) with quick onset of action such as formoterol; and (iii) short-acting anti-muscarinic agents (SAMA) such as ipratropium. SABA are traditionally the most common agents used for immediate symptomatic relief by patients with asthma. Formoterol is equally effective in this regard. [7] However, its use as a rescue medication is generally discouraged due to concerns related to LABA monotherapy in asthma. SAMA are generally less preferred as their onset of action is delayed as compared to SABA, although direct comparisons between the two agents have not been performed. A Cochrane review also noted that although SAMA was superior to placebo, a combination of SABA plus SAMA was not better than SABA alone. [8]
Oral beta-agonists (salbutamol, terbutaline) are also commonly used as reliever medications in India because these are less expensive. However, they have an unfavourable risk-benefit ratio and cannot be recommended for routine use.
Inhaled Corticosteroids (ICS)
ICS are the most potent and consistently effective long-term controller medication for asthma. ICS suppress airway inflammation, thereby improving asthma control. Therefore, they are uniformly recommended by all major guidelines as the first-line controller agent for managing stable asthma. Five molecules are currently marketed in India: beclomethasone, budesonide, fluticasone, ciclesonide and mometasone. Although these agents differ slightly in their pharmacokinetic and pharmacodynamic properties, all have largely similar efficacy and safety profile when used in equivalent doses. [9] ,[10] ,[11]
All ICS are clearly superior to placebo in reducing severity of symptoms, improving pulmonary function, improving asthma control and quality of life, and reducing the need for reliever medication, exacerbations, emergency visits and hospitalizations. [12] ,[13] ,[14] ,[15] ICS monotherapy has also been shown to be superior to LABA monotherapy in reducing inflammatory markers, treatment failures and asthma exacerbations in the SOCS (Salmeterol Or Corticosteroids Study) trial. [16] Another Cochrane review showed that ICS monotherapy was better than treatment with anti-leukotriene agents in improving symptoms, pulmonary function and quality of life, and decreasing the use of rescue medication and asthma exacerbations. [17] ICS monotherapy has also been shown to be superior to oral methylxanthine therapy in six randomized controlled trials. [18] ,[19] ,[20] ,[21] ,[22] ,[23] Thus, current evidence clearly supports the use of ICS as the primary controller agent for managing patients with stable asthma.
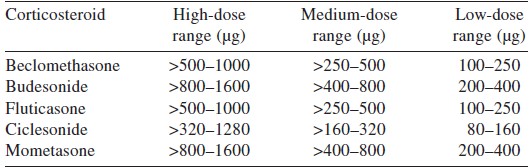
There has been much debate on the optimum ICS dosing. While low doses may fail to achieve adequate disease control, higher doses may be associated with adverse effects. At high doses, ICS use can manifest with systemic side-effects, the most important of which is suppression of the hypothalamo-pituitary-adrenal axis at higher doses. Other complications include eye problems such as cataract and glaucoma, skin bruising, reduction in bone mineral density, growth retardation in children, and most importantly an increased risk of infections. In addition, local oropharyngeal deposition can lead to throat irritation, cough, and sometimes even dysphonia or oral candidiasis. However, evidence suggests that bulk of the clinical improvement observed with ICS occurs at low-to-moderate doses ([Table - 1]), beyond which further dose increments provide negligible additional benefit. [24] ,[25] ,[26] ,[27] ,[28] Also, initiating ICS therapy at low-dose may be as effective as using an initial higher dose and later stepping down. [29] This is particularly important in minimizing local and systemic side-effects of ICS therapy. Another recently proposed shift in dosing strategy is relevant for patients with mild persistent asthma. These patients may receive intermittent ICS therapy on an as-needed basis rather than as regular daily therapy, because studies have shown that the former is not inferior to the latter. [30] ,[31] ,[32] ,[33] Such an approach may not work for patients with moderate disease. [34]
Inhaled Long-Acting Beta-Agonists (LABA)
Three potent selective LABA are currently marketed in India: salmeterol, formoterol and indacaterol. The last one is an ultra-long-acting agent suitable for once daily dosing. Both formoterol and salmeterol need to be given twice daily. However, formoterol has a more rapid onset of action, and can also be used as a rescue medication. As compared to placebo, salmeterol and formoterol significantly improve pulmonary function, symptoms and quality of life, and decrease the use of rescue medications and exacerbation of asthma. [35] Not much data are available on the use of indacaterol in managing asthma.
Since LABA have no effect on modulating airway inflammation, their use as monotherapy is discouraged in asthma. LABA are excellent bronchodilators and lead to substantial symptomatic improvement. However, they allow airway inflammation to progress unchecked subclinically, and this may lead to poorer long-term outcomes. The Serevent National Surveillance study reported that salmeterol monotherapy was associated with more life-threatening experiences and asthma-related deaths. [36] Other studies have also shown that switching patients from ICS monotherapy to LABA monotherapy results in a loss of disease control. A recent meta-analysis also concluded that monotherapy with salmeterol increased the risk of asthma-related mortality, which was reduced with concurrent ICS use. [37]
Combined ICS And LABA
Since LABA monotherapy is considered improper, LABA are now predominantly used as add-on agents to ICS. Several large trials such as OPTIMA (Oxis and Pulmicort Turbuhaler in Management of Asthma), FACET (Formoterol and Corticosteroid Establishing Trial) and GOAL (Gaining Optimal Asthma controL) have confirmed that this combination further improves asthma control and reduces the risk of exacerbations. [38] ,[39] ,[40] The SLIC (SaLmeterol with or without Inhaled Corticosteroids) study also showed that in patients poorly controlled on ICS monotherapy, addition of LABA allowed reduction of ICS dose by half. [41] A Cochrane review of 71 studies concluded that the risk of exacerbations requiring oral corticosteroids could be reduced by 28% by adding LABA to ICS, without any increase in side-effects. [42] Further, among patients with asthma uncontrolled on low ICS doses, adding LABA is better than increasing ICS dose in improving asthma symptoms and pulmonary function, and decreasing exacerbations and steroid-related adverse effects. [43] The ICS-LABA combination works best in reducing exacerbations among all long-term treatment strategies for asthma. [44]
Fixed-dose ICS-LABA combinations are now commonly available for use as maintenance therapy in asthma. For several years, additional reliever medications were suggested as supplements on an as-needed basis during periods of worsening asthma control immediately before an asthma exacerbation. It has, however, recently been proposed that an additional dose of ICS in this phase augments the anti-inflammatory action, and may help in aborting an impending exacerbation of asthma. This has evolved into the single inhaler therapy (or SiT) approach, using a combination of fast-acting LABA (formoterol) with ICS that can be successfully used as both maintenance and reliever medication in routine clinical practice. [45] Most studies comparing SiT with conventional therapy have used formoterol-budesonide turbuhaler, and have proposed SMART (single agent for maintenance and reliever therapy) as an alternative approach. However, any ICS and any ′good′ inhalation device can be used in the SiT approach. Studies comparing SiT with ICS monotherapy have reported better asthma symptoms, disease control and pulmonary function, with longer time to first exacerbation. [46] ,[47] ,[48] ,[49] Most studies comparing SiT with fixed-dose ICS-LABA combinations have also shown similar results. [49] ,[50] ,[51] ,[52] ,[53] ,[54] ,[55] ,[56] ,[57] ,[58] Other studies comparing the SiT approach with conventional guideline-based best practice have shown better asthma control, but similar lung function and exacerbations of asthma. [59] ,[60] ,[61] ,[62] ,[63] An open-labelled observational study that also recruited Indian patients showed better asthma control with SiT. [64] Notably, the total cumulative dose of ICS in most of these studies was lesser with the SiT approach, probably because equivalent asthma control could be achieved at an overall lower daily ICS maintenance dose. Few meta-analyses have concluded that the SiT strategy statistically significantly reduces exacerbations of asthma requiring oral corticosteroids. [65] ,[66] ,[67]
Anti-Leukotriene Agents
Cysteinyl leukotrienes, generated through the 5-lipoxygenase pathway of arachidonic acid metabolism, are potent inflammatory mediators in asthma. However, their effects cannot be blocked by corticosteroids, and hence anti-leukotriene agents with complementary anti-inflammatory modes of action have been developed. [68] Oral montelukast, a cysteinyl leukotriene-1 receptor antagonist (LTRA), is the most commonly used antileukotriene agent in asthma. A major advantage of montelukast is that it can be administered orally and is well tolerated.
LTRAs can be used alone in patients with mild asthma, or as add-on to ICS or ICS-LABA therapy. [69] When used as a single agent in patients with mild-to-moderate asthma, LTRAs are inferior to ICS in providing symptomatic relief or preventing acute exacerbations. [17] ,[70] However, in a real-world scenario, montelukast and ICS monotherapy appear equivalent for patients with mild asthma. [71] Large observational studies have shown that addition of LTRA improves asthma control and quality of life in poorly controlled patients while receiving ICS alone or an ICS-LABA combination. [72] ,[73] ,[74] ,[75] A recent Cochrane review concluded that addition of LTRAs to ICS therapy leads to a non-significant risk reduction for exacerbations of asthma requiring oral corticosteroids. [76] Another Cochrane review has shown that an ICS-LTRA combination was inferior to an ICS-LABA combination in improving lung function or preventing exacerbations of asthma. [77]
Long-Acting Anti-Muscarinic Agents
Tiotropium, a long-acting anti-muscarinic agent (LAMA), is widely used as first-line therapy in managing patients with chronic obstructive pulmonary disease. There is now increasing interest in the use of tiotropium as an additional agent for managing patients inadequately controlled with ICS or ICS-LABA therapy. [78] The clinical benefit from addition of tiotropium to ICS therapy in poorly controlled patients has been shown to be similar to that of addition of LABA. [79] ,[80] ,[81] However, when added to an ongoing treatment with an ICS-LABA combination, tiotropium leads to better pulmonary function and fewer exacerbations. [82] ,[83] ,[84] Hence, tiotropium can be used as an additional drug for poorly controlled patients despite moderate-to-high dose ICS-LABA combination therapy, or in those having remodelled phenotype of asthma.
Methylxanthines
Theophylline (dimethylxanthine) continues to be commonly prescribed to patients with asthma in India as it is cheap and easily available. The weak bronchodilator action of theophylline, observed at blood levels of 10-20 mg/L after a standard dose, is linked to adenosine receptor antagonism and non-specific phosphodiesterase inhibition. On the other hand, at lower doses, resulting in blood levels of only 5-10 mg/L, theophylline manifests anti-inflammatory action resulting from histone deacetylase activation. [85] At these low doses, the safety profile of theophylline is also much better, and hence this appears to be the current preferred dose.
Theophylline monotherapy has been shown to be inferior to ICS monotherapy in improving clinical outcomes in asthma. [18] ,[19] ,[20] ,[21] ,[22] ,[23] Two meta-analyses have also shown LABA monotherapy to be superior to theophylline monotherapy in improving symptoms and pulmonary function. [86] ,[87] Hence, theophylline appears inferior to both ICS and LABA in managing asthma. However, addition of theophylline to low-dose ICS may be as good as doubling the ICS dose. [88] ,[89] ,[90] ,[91] Among patients with asthma who are poorly controlled with ICS-LABA therapy, adding theophylline can further improve pulmonary function and reduce exacerbations. [92] ,[93]
Doxofylline is a newer alternative to theophylline which is postulated to have fewer side-effects due to lack of adenosine receptor antagonism and calcium channel receptor blocking ability. [94] Studies comparing standard dose theophylline and doxofylline have shown the drugs to be equally effective, with doxofylline associated with fewer adverse events. [95] ,[96] There are no data comparing low-dose theophylline with doxofylline in asthma.
Monoclonal Antibody Against Immunoglobulin E
Omalizumab is a recombinant humanized immunoglobulin G1 monoclonal antibody that binds immunoglobulin E (IgE) with high affinity. Meta-analyses have shown that it reduced ICS dose requirement and frequency of asthma exacerbations when used as an adjunct to ICS in patients with moderate-to-severe asthma. [97] ,[98] Similar benefits were not observed among patients receiving oral steroids. Most of these trials evaluated omalizumab in patients with elevated serum IgE levels and a positive skin test to aero-allergens, and the drug is currently reserved for this subset of patients if they respond inadequately to conventional treatment. These studies in general showed that a favourable response to omalizumab therapy occurred within 12 weeks. [99] Hence, omalizumab should be administered for at least 12-16 weeks before evaluating clinical response. The dose of omalizumab is determined by body weight and serum IgE levels (0.016 mg/kg per IU/mL of IgE per month), and is administered subcutaneously every 2 to 4 weeks. Only 30%-50% of patients show some response, and the total duration of treatment is not known. [100] Few experts believe that it may be given lifelong, as discontinuing therapy can lead to severe rebound exacerbations. [101] There is a theoretical risk of parasitic infections or malignancy with long-term use of omalizumab, and more safety data are needed before its routine use can be recommended.
Route Of Administration And Devices
The major classes of drugs useful in managing a patient with stable asthma (corticosteroids and beta-agonists) are available both in oral and inhalational formulations. Wherever possible, the inhaled route is clearly superior to the oral route. Ingested drugs must get absorbed into the systemic circulation first, and only a small fraction finally reaches the lungs. Inhaled drugs, on the other hand, are directly delivered to their site of intended action, and therefore need only a small fraction of the oral dose for equivalent effect. For this reason, inhalational agents are associated with fewer systemic adverse effects. Except for special situations such as severe financial constraints, non-availability of inhaled drugs, etc., there is currently no indication to prescribe oral medications.
The inhaled drugs can be administered through pressurized metered dose inhalers (pMDI), dry powder inhalers (DPI) or nebulizers. In general, pMDIs are the best standardized devices with fractional drug delivery to the lungs in a well-defined range. However, a major issue with pMDI use is the hand-mouth coordination. Studies have shown that a substantial proportion of patients using pMDIs do so incorrectly. [102] ,[103] Therefore, it is important to teach patients how to correctly use their pMDI, and verify their technique at each visit. For patients who cannot coordinate their inhaler well, one can suggest using a valved spacer chamber in conjunction with the pMDI. This not only removes the problems of coordination, but also reduces oral drug deposition and improves drug delivery to lungs. Alternatively, patients can use a nebulizer for routine use. Contrary to popular belief, drug delivery to lungs through a nebulizer is similar to that achieved when using pMDI with a spacer device. [104] DPIs are a heterogeneous collection of breath-actuated devices with highly variable inspiratory airflow requirements and drug delivery to lungs. Most DPIs are inferior to pMDIs as they generate larger particle size for inhalation, and hence cause more oropharyngeal drug deposition and lesser drug delivery to the lungs. On a long-term basis, they are also costlier than pMDIs. However, several patients find DPIs easier to use. Ultimately, one must strike a balance between efficacy (in terms of drug delivery to lungs at a particular unit dose) and patient preference while selecting the device for inhaled drugs. Whatever be the device, patients must be adequately educated about the proper technique of usage and maintenance of the device, and the technique must be checked at each follow-up visit.
General Approach To Management
A stepwise approach is considered practical for managing patients with stable asthma ([Table - 2]). The decision to treat in any step is based on severity of symptoms as well as response to already prescribed drugs ([Figure - 1]).
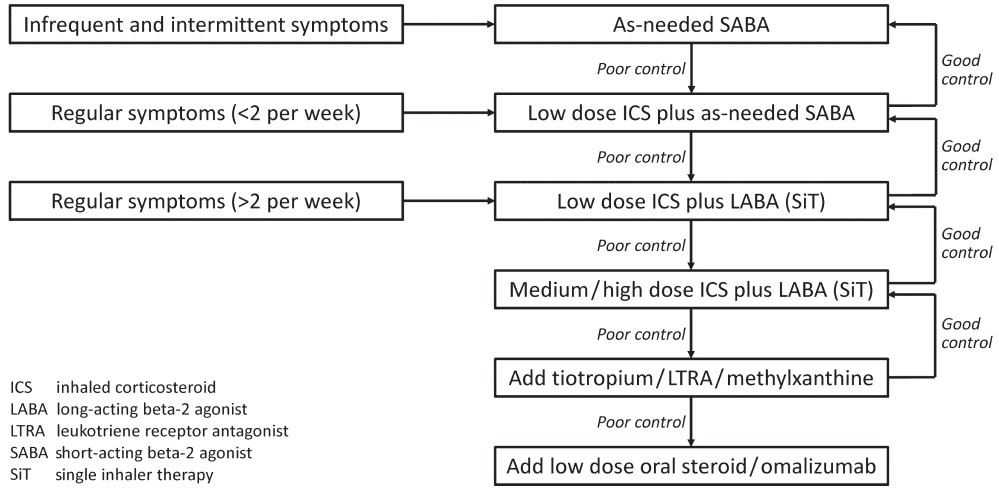 |
| Figure 1. A suggested algorithm to initiate and modulate asthma pharmacotherapy |

Step 1: As-needed reliever
The lowest step in asthma management is useful only for those patients who experience intermittent symptoms (typically less than twice a month), and are symptom-free between these episodes. These patients can be managed with a reliever medication alone, taken whenever required. A SABA is the preferred agent for these patients.
Step 2: Single controller
Patients having more frequent or regular symptoms (typically between twice a month and twice a week) should not be managed with reliever drugs alone. They should take a controller medication in addition to the reliever medication on an as-needed basis. Low dose ICS is preferred as the controller drug, although LTRA monotherapy can be prescribed as an alternative to those who cannot take ICS. Monotherapy with either methylxanthine or LABA is discouraged.
Step 3: Dual controllers
If asthma remains poorly controlled despite low dose ICS (step 2), or if symptoms occur more than twice a week or during night in treatment-naïve patients, therapy needs to be augmented. The preferred approach is addition of a LABA to the low-dose ICS which the patient is already taking. Other potential alternatives include doubling the ICS dose or combining a LTRA or theophylline with ICS. However, the options are not as efficacious as adding LABA to ICS. The SiT approach, using a formoterol-based combination, should always be preferred in such (and subsequent) settings.
Step 4: Two or more controllers
This is a complex treatment step for patients uncontrolled despite step 3 therapy, and represents all the sequential increases in medication before a patient is considered a candidate for oral corticosteroid or other therapy.
The preferred therapeutic option for these patients is continuing with an ICS-LABA combination and hiking up the dose of ICS to medium and high doses sequentially. Most patients will not derive additional benefit from increments in ICS dosing beyond the medium dose range. High-dose ICS may be tried for 3 months, and de-escalated if there is no clinical improvement. If a patient was not taking a LABA at step 3, it must be added before any further change in therapy.
In case patients remain uncontrolled despite a moderate-to-high dose ICS-LABA combination, tiotropium, LTRA, and/or methylxanthine can be added. This treatment is highly individualized, based on preferences of patients and physicians, and availability, tolerability and cost of drugs.
Step 5: Other add-on drugs
For patients who fail to respond to all the treatment approaches mentioned above (sometimes referred to as difficult-to-treat asthma), a trial of oral corticosteroids, or other targeted therapies such as omalizumab, may be considered. Although no clinical trial has specifically studied the role of oral corticosteroids among patients with difficult-to-control asthma, they can be considered in such a scenario at the lowest possible dose and for the shortest possible duration required to achieve asthma control in view of their many adverse effects.
Monitoring And Modulation Of Therapy
The optimal frequency of follow-up visit of patients with asthma is between 1 and 3 months, although it may be more or less depending on an individual clinical scenario. Patients should be assessed for asthma control at each visit, and categorized as having either adequate or poor control of asthma. A patient is considered uncontrolled if she/he has either (i) daytime symptoms or rescue medication use more than twice per week; (ii) nocturnal symptoms or awakening; (iii) limitation of daily activities; or (iv) diminished peak expiratory flow (<80% of predicted best). Patients not having any of these features may be considered well controlled. At each visit, it is also important to enquire about compliance to therapy and any treatment-related adverse effects. An effort should be made at each visit to verify that the patient is indeed taking the inhaled medications using appropriate technique and device.
Therapy needs to be stepped up for patients with poorly controlled asthma on their ongoing treatment ([Figure - 1]). The best time to make such a decision is not clearly defined. A subgroup analysis of data from the GOAL study showed that each individual item used to assess asthma control took a different time to exhibit control. Improvement in peak expiratory flow or nocturnal symptoms happened much more quickly than other symptoms. In general, daytime symptoms took the longest to improve. [105] Overall, stepping up therapy may be considered at intervals of 1-3 months if a patient remains uncontrolled despite using prescribed medications correctly, although such titration of therapy needs to be individualized. For patients using an ICS-LABA combination (in particular those using SiT), dosage increments should be done after ensuring that the LABA dose stays within the safe therapeutic range.
For those who are adequately controlled on treatment, one can attempt stepping down treatment with an aim to maintain control at lowest possible drug doses ([Figure - 1]). The optimal timing and sequence of stepping down however remains controversial. [106] ,[107] In general, stepping down treatment should be considered only after 2-3 months of good asthma control. For patients with asthma known to have seasonal exacerbations, such modifications should not be attempted during the time disease control is likely to be poor. All patients whose drugs are reduced should be closely monitored to detect subsequent worsening of disease control. For patients taking more than two controller drugs, the non-ICS, non-LABA controller drugs should be initially reduced/discontinued. Patients well controlled on medium or high-dose ICS, with or without LABA, can decrease/halve their ICS dose every three months till they attain a low ICS dose. Data suggest that patients controlled on high-dose ICS can be switched to a lower dose without losing disease control. [108] ,[109] ,[110] When stepping down from ICS-LABA combination therapy, it has also been shown that decreasing ICS dose is better than discontinuing LABA in maintaining asthma control. [111] ,[112] ,[113] Discontinuing LABA might be associated with higher likelihood of exacerbations requiring definitive treatment. Once a patient is well controlled on a low-dose ICS and LABA combination, one can attempt discontinuing LABA altogether. However, there is a possibility of losing asthma control and patients should be carefully monitored for the same. [114] ,[115] ,[116] ,[117] When low-dose ICS monotherapy is sufficient to maintain good control, patients can try reducing the frequency of daily dosing. A meta-analysis of nine studies has reported that budesonide given once or twice daily is equally effective. [118] Finally, if a patient remains controlled even on such low ICS doses, one can try stopping regular ICS (step 1). This may, however, increase the risk of exacerbations. [119] Alternatively, one can switch to as-needed ICS, although there is insufficient evidence regarding such a choice. [120]
Conclusion
Several classes of inhaled and systemic drugs are available for long-term medical management of patients with asthma. A typical asthma prescription should be tailored to the patient′s needs and preferences. Inhaled drugs are preferred to oral agents wherever feasible. ICS are the mainstay of therapy for controlling airway inflammation, and LABA are useful for patients not adequately controlled with ICS alone. Clinicians should build prescriptions based on treatment recommendations from evidence-based guidelines available for Indian patients.
| 1. | Global Initiative for Asthma (GINA). Global Strategy for Asthma Management and Prevention; 2015. Available at www.ginasthma.org/ (accessed on 10 Nov 2016) [Google Scholar] |
| 2. | British Thoracic Society; Scottish Intercollegiate Guidelines Network. British guideline on the management of asthma. Thorax 2014; 69 Suppl 1:1-192. [Google Scholar] |
| 3. | Ohta K, Ichinose M, Nagase H, Yamaguchi M, Sugiura H, Tohda Y, et al.; Japanese Society of Allergology. Japanese Guideline for Adult Asthma 2014. Allergol Int 2014; 63: 293-333. [Google Scholar] |
| 4. | Lougheed MD, Leniere C, Ducharme FM, Licskai C, Dell SD, Rowe BH, et al.; Canadian Thoracic Society Asthma Clinical Assembly. Canadian Thoracic Society 2012 guideline update: Diagnosis and management of asthma in preschoolers, children and adults: Executive summary. Can Respir J 2012; 19: e81-8. [Google Scholar] |
| 5. | Jindal SK, Gupta D, Aggarwal AN, Agarwal R; World Health Organization; Government of India. Guidelines for management of asthma at primary and secondary levels of health care in India. Indian J Chest Dis Allied Sci 2005; 47: 309-43. [Google Scholar] |
| 6. | Agarwal R, Dhooria S, Aggarwal AN, Maturu VN, Sehgal IS, Muthu V, et al. Guidelines for diagnosis and management of bronchial asthma: Joint ICS/NCCP (I) recommendations. Lung India 2015; 32 (suppl 1):S3-S42. [Google Scholar] |
| 7. | Welsh EJ, Cates CJ. Formoterol versus short-acting beta agonists as relief medication for adults and children with asthma. Cochrane Database Syst Rev 2010; (9): CD008418. [Google Scholar] |
| 8. | Westby M, Benson M, Gibson P. Anticholinergic agents for chronic asthma in adults. Cochrane Database Syst Rev 2004; (3): CD003269. [Google Scholar] |
| 9. | Adams N, Bestall JM, Jones PW. Inhaled beclomethasone versus budesonide for chronic asthma. Cochrane Database Syst Rev 2002; (1): CD003530. [Google Scholar] |
| 10. | Adams N, Lasserson TJ, Cates CJ, Jones PW. Fluticasone versus beclomethasone or budesonide for chronic asthma in adults and children. Cochrane Database Syst Rev 2007; (4): CD002310. [Google Scholar] |
| 11. | Manning P, Gibson PG, Lasserson TJ. Ciclesonide versus other inhaled steroids for chronic asthma in children and adults. Cochrane Database Syst Rev 2008; (2): CD007031. [Google Scholar] |
| 12. | Adams N, Bestall J, Jones PW. Budesonide for chronic asthma in children and adults. Cochrane Database Syst Rev 2001; (4): CD003274. [Google Scholar] |
| 13. | Adams NP, Bestall JC, Lasserson TJ, Jones P, Cates CJ. Fluticasone versus placebo for chronic asthma in adults and children. Cochrane Database Syst Rev 2008; (4): CD003135. [Google Scholar] |
| 14. | Adams NP, Bestall JB, Malouf R, Lasserson TJ, Jones PW. Inhaled beclomethasone versus placebo for chronic asthma. Cochrane Database Syst Rev 2005; (1): CD002738. [Google Scholar] |
| 15. | Manning P, Gibson PG, Lasserson TJ. Ciclesonide versus placebo for chronic asthma in adults and children. Cochrane Database Syst Rev 2008 (2): CD006217. [Google Scholar] |
| 16. | Lazarus SC, Boushey HA, Fahy JV, Chinchilli VM, Lemanske RF Jr, Sorkness CA, et al. Asthma Clinical Research Network for the National Heart, Lung, and Blood Institute. Long-acting beta2-agonist monotherapy vs continued therapy with inhaled corticosteroids in patients with persistent asthma: A randomized controlled trial. JAMA 2001; 285: 2583-93. [Google Scholar] |
| 17. | Chauhan BF, Ducharme FM. Anti leukotriene agents compared to inhaled corticosteroids in the management of recurrent and/or chronic asthma in adults and children. Cochrane Database Syst Rev 2012; (5): CD002314. [Google Scholar] |
| 18. | Youngchaiyud P, Permpikul C, Suthamsmai T, Wong E. A double-blind comparison of inhaled budesonide, long-acting theophylline, and their combination in treatment of nocturnal asthma. Allergy 1995; 50: 28-33. [Google Scholar] |
| 19. | Reed CE, Offord KP, Nelson HS, Li JT, Tinkelman DG. Aerosol beclomethasone dipropionate spray compared with theophylline as primary treatment for chronic mild-to-moderate asthma. The American Academy of Allergy, Asthma and Immunology Beclomethasone Dipropionate-Theophylline Study Group. J Allergy Clin Immunol 1998; 101: 14-23. [Google Scholar] |
| 20. | Morali T, Yilmaz A, Erkan F, Akkaya E, Ece F, Baran R. Efficacy of inhaled budesonide and oral theophylline in asthmatic subjects. J Asthma 2001; 38: 673-9. [Google Scholar] |
| 21. | Tinkelman DG, Reed CE, Nelson HS, Offord KP. Aerosol beclomethasone dipropionate compared with theophylline as primary treatment of chronic, mild to moderately severe asthma in children. Pediatrics1993; 92: 64-77. [Google Scholar] |
| 22. | Dahl R, Larsen BB, Venge P. Effect of long-term treatment with inhaled budesonide or theophylline on lung function, airway reactivity and asthma symptoms. Respir Med 2002; 96: 432-8. [Google Scholar] |
| 23. | Yurdakul AS, Taci N, Eren A, Sipit T. Comparative efficacy of once-daily therapy with inhaled corticosteroid, leukotriene antagonist or sustained-release theophylline in patients with mild persistent asthma. Respir Med 2003; 97: 1313-19. [Google Scholar] |
| 24. | Szefler SJ, Martin RJ, King TS, Boushey HA, Cherniack RM, Chinchilli VM, et al.; Asthma Clinical Research Network of the National Heart Lung, and Blood Institute. Significant variability in response to inhaled corticosteroids for persistent asthma. J Allergy Clin Immunol 2002; 109: 410-18. [Google Scholar] |
| 25. | Holt S, Suder A, Weatherall M, Cheng S, Shirtcliffe P, Beasley R. Dose-response relation of inhaled fluticasone propionate in adolescents and adults with asthma: Meta-analysis. BMJ 2001; 323: 253-6. [Google Scholar] |
| 26. | Masoli M, Weatherall M, Holt S, Beasley R. Clinical dose-response relationship of fluticasone propionate in adults with asthma. Thorax 2004; 59: 16-20. [Google Scholar] |
| 27. | Adams NP, Bestall JC, Jones P, Lasserson TJ, Griffiths B, Cates CJ. Fluticasone at different doses for chronic asthma in adults and children. Cochrane Database Syst Rev 2008; (4): CD003534. [Google Scholar] |
| 28. | Adams N, Bestall J, Jones PW. Budesonide at different doses for chronic asthma. Cochrane Database Syst Rev 2001 (4): Cd003271. [Google Scholar] |
| 29. | Powell H, Gibson PG. High dose versus low dose inhaled corticosteroid as initial starting dose for asthma in adults and children. Cochrane Database Syst Rev 2004; (2): CD004109. [Google Scholar] |
| 30. | Calhoun WJ, Ameredes BT, King TS, Icitovic N, Bleecker ER, Castro M, et al. Asthma Clinical Research Network of the National Heart, Lung, and Blood Institute. Comparison of physician-, biomarker-, and symptom-based strategies for adjustment of inhaled corticosteroid therapy in adults with asthma: The BASALT randomized controlled trial. JAMA 2012; 308: 987-97. [Google Scholar] |
| 31. | Papi A, Canonica GW, Maestrelli P, Paggiaro P, Olivieri D, Pozzi E, et al. BEST Study Group. Rescue use of beclomethasone and albuterol in a single inhaler for mild asthma. N Engl J Med 2007; 356: 2040-52. [Google Scholar] |
| 32. | Boushey HA, Sorkness CA, King TS, Sullivan SD, Fahy JV, Lazarus SC, et al. National Heart, Lung, and Blood Institute′s Asthma Clinical Research Network. Daily versus as-needed corticosteroids for mild persistent asthma. N Engl J Med 2005; 352: 1519-28. [Google Scholar] |
| 33. | Chauhan BF, Chartrand C, Ducharme FM. Intermittent versus daily inhaled corticosteroids for persistent asthma in children and adults. Cochrane Database Syst Rev 2013; 2: CD009611. [Google Scholar] |
| 34. | Papi A, Marku B, Scichilone N, Maestrelli P, Paggiaro P, Saetta M, et al. AIFASMA Study Group. Regular versus as-needed budesonide and formoterol combination treatment for moderate asthma: A non-inferiority, randomised, double-blind clinical trial. Lancet Respir Med 2015; 3: 109-19. [Google Scholar] |
| 35. | Walters EH, Gibson PG, Lasserson TJ, Walters JA. Long-acting beta2-agonists for chronic asthma in adults and children where background therapy contains varied or no inhaled corticosteroid. Cochrane Database Syst Rev 2007; (1): CD001385. [Google Scholar] |
| 36. | Castle W, Fuller R, Hall J, Palmer J. Serevent nationwide surveillance study: Comparison of salmeterol with salbutamol in asthmatic patients who require regular bronchodilator treatment. BMJ 1993; 306: 1034-7. [Google Scholar] |
| 37. | Weatherall M, Wijesinghe M, Perrin K, Harwood M, Beasley R. Meta-analysis of the risk of mortality with salmeterol and the effect of concomitant inhaled corticosteroid therapy. Thorax 2010; 65: 39-43. [Google Scholar] |
| 38. | O′Byrne PM, Barnes PJ, Rodriguez-Roisin R, Runnerstrom E, Sandstrom T, Svensson K, et al. Low dose inhaled budesonide and formoterol in mild persistent asthma: The OPTIMA randomized trial. Am J Respir Crit Care Med 2001; 164: 1392-7. [Google Scholar] |
| 39. | Pauwels RA, Löfdahl CG, Postma DS, Tattersfield AE, O′Byrne P, Barnes PJ, et al. Effect of inhaled formoterol and budesonide on exacerbations of asthma. Formoterol and Corticosteroids Establishing Therapy (FACET) International Study Group. N Engl J Med 1997; 337: 1405-11. [Google Scholar] |
| 40. | Bateman ED, Boushey HA, Bousquet J, Busse WW, Clark TJ, Pauwels RA, et al. GOAL Investigators Group. Can guideline-defined asthma control be achieved? The Gaining Optimal Asthma Control study. Am J Respir Crit Care Med 2004; 170: 836-44. [Google Scholar] |
| 41. | Lemanske RF Jr, Sorkness CA, Mauger EA, Lazarus SC, Boushey HA, Fahy JV, et al. Asthma Clinical Research Network for the National Heart, Lung, and Blood Institute. Inhaled corticosteroid reduction and elimination in patients with persistent asthma receiving salmeterol: A randomized controlled trial. JAMA 2001; 285: 2594-603. [Google Scholar] |
| 42. | Ducharme FM, Ni Chroinin M, Greenstone I, Lasserson TJ. Addition of long-acting beta 2-agonists to inhaled corticosteroids versus same dose inhaled corticosteroids for chronic asthma in adults and children. Cochrane Database Syst Rev 2010; (5): CD005535. [Google Scholar] |
| 43. | Ducharme FM, Ni Chroinin M, Greenstone I, Lasserson TJ. Addition of long-acting beta 2-agonists to inhaled steroids versus higher dose inhaled steroids in adults and children with persistent asthma. Cochrane Database Syst Rev 2010; (4): CD005533. [Google Scholar] |
| 44. | Loymans RJ, Gemperli A, Cohen J, Rubinstein SM, Sterk PJ, Reddel HK, et al. Comparative effectiveness of long term drug treatment strategies to prevent asthma exacerbations: Network meta-analysis. BMJ 2014; 348: 3009. [Google Scholar] |
| 45. | Ställberg B, Naya I, Ekelund J, Eckerwall G. Real-life use of budesonide/formoterol in clinical practice: A 12-month follow-up assessment in a multi-national study of asthma patients established on single-inhaler maintenance and reliever therapy. Int J Clin Pharmacol Ther 2015; 53: 447-55. [Google Scholar] |
| 46. | Sovani MP, Whale CI, Oborne J, Cooper S, Mortimer K, Ekström T, et al. Poor adherence with inhaled corticosteroids for asthma: Can using a single inhaler containing budesonide and formoterol help? Br J Gen Pract 2008; 58: 37-43. [Google Scholar] |
| 47. | Scicchitano R, Aalbers R, Ukena D, Manjra A, Fouquert L, Centanni S, et al. Efficacy and safety of budesonide/formoterol single inhaler therapy versus a higher dose of budesonide in moderate to severe asthma. Curr Med Res Opin 2004; 20: 1403-18. [Google Scholar] |
| 48. | Rabe KF, Pizzichini E, Ställberg B, Romero S, Balanzat AM, Atienza T, et al. Budesonide/formoterol in a single inhaler for maintenance and relief in mild-to-moderate asthma: A randomized, double-blind trial. Chest 2006; 129: 246-56. [Google Scholar] |
| 49. | O′Byrne PM, Bisgaard H, Godard PP, Pistolesi M, Palmqvist M, Zhu Y, et al. Budesonide/formoterol combination therapy as both maintenance and reliever medication in asthma. Am J Respir Crit Care Med 2005; 171: 129-36. [Google Scholar] |
| 50. | Vogelmeier C, D′Urzo A, Pauwels R, Merino JM, Jaspal M, Boutet S, et al. Budesonide/formoterol maintenance and reliever therapy: An effective asthma treatment option? Eur Respir J 2005; 26: 819-28. [Google Scholar] |
| 51. | Rabe KF, Atienza T, Magyar P, Larsson P, Jorup C, Lalloo UG. Effect of budesonide in combination with formoterol for reliever therapy in asthma exacerbations: A randomised controlled, double-blind study. Lancet 2006; 368: 744-53. [Google Scholar] |
| 52. | Ställberg B, Ekström T, Neij F, Olsson P, Skoogh BE, Wennergren G, et al.; SHARE trial group. A real-life cost-effectiveness evaluation of budesonide/formoterol maintenance and reliever therapy in asthma. Respir Med 2008; 102: 1360-70. [Google Scholar] |
| 53. | Lundborg M, Wille S, Bjermer L, Tilling B, Lundgren M, Telg G, et al. Maintenance plus reliever budesonide/formoterol compared with a higher maintenance dose of budesonide/formoterol plus formoterol as reliever in asthma: An efficacy and cost-effectiveness study. Curr Med Res Opin 2006; 22: 809-21. [Google Scholar] |
| 54. | Kuna P, Peters MJ, Manjra AI, Jorup C, Naya IP, Martínez-Jimenez NE, et al. Effect of budesonide/formoterol maintenance and reliever therapy on asthma exacerbations. Int J Clin Pract 2007; 61: 725-36. [Google Scholar] |
| 55. | Bousquet J, Boulet LP, Peters MJ, Magnussen H, Quiralte J, Martinez-Aguilar NE, et al. Budesonide/formoterol for maintenance and relief in uncontrolled asthma vs. high-dose salmeterol/fluticasone. Respir Med 2007; 101: 2437-46. [Google Scholar] |
| 56. | Patel M, Pilcher J, Beasley R. Combination ICS/fast-onset LABA inhaler as maintenance and reliever therapy: The future for uncontrolled adult asthma? Expert Rev Respir Med 2013; 7: 451-4. [Google Scholar] |
| 57. | Papi A, Corradi M, Pigeon-Francisco C, Baronio R, Siergiejko Z, Petruzzelli S, et al. Beclometasone-formoterol as maintenance and reliever treatment in patients with asthma: A double-blind, randomised controlled trial. Lancet Respir Med 2013; 1: 23-31. [Google Scholar] |
| 58. | Patel M, Pilcher J, Pritchard A, Perrin K, Travers J, Shaw D, et al.; SMART Study Group. Efficacy and safety of maintenance and reliever combination budesonide-formoterol inhaler in patients with asthma at risk of severe exacerbations: A randomised controlled trial. Lancet Respir Med 2013; 1: 32-42. [Google Scholar] |
| 59. | Sears MR, Boulet LP, Laviolette M, Fitzgerald JM, Bai TR, Kaplan A, et al. Budesonide/formoterol maintenance and reliever therapy: Impact on airway inflammation in asthma. Eur Respir J 2008; 31: 982-9. [Google Scholar] |
| 60. | Riemersma RA, Postma D, van der Molen T. Budesonide/formoterol maintenance and reliever therapy in primary care asthma management: Effects on bronchial hyperresponsiveness and asthma control. Prim Care Respir J 2012; 21: 50-6. [Google Scholar] |
| 61. | Søes-Petersen U, Kava T, Dahle R, Lei Y, Dam N. Budesonide/formoterol maintenance and reliever therapy versus conventional best standard treatment in asthma in an attempted ′real life′ setting. Clin Respir J 2011; 5: 173-82. [Google Scholar] |
| 62. | Louis R, Joos G, Michils A, Vandenhoven G. A comparison of budesonide/formoterol maintenance and reliever therapy vs. conventional best practice in asthma management. Int J Clin Pract 2009; 63: 1479-88. [Google Scholar] |
| 63. | Demoly P, Louis R, Søes-Petersen U, Naya I, Carlsheimer A, Worth H, et al. Budesonide/formoterol maintenance and reliever therapy versus conventional best practice. Respir Med 2009; 103: 1623-32. [Google Scholar] |
| 64. | Zhong N, Lin J, Mehta P, Ngamjanyaporn P, Wu TC, Yunus F. Real-life effectiveness of budesonide/formoterol maintenance and reliever therapy in asthma patients across Asia: SMARTASIA study. BMC Pulm Med 2013; 13: 22. [Google Scholar] |
| 65. | Agarwal R, Khan A, Aggarwal AN, Gupta D. Is the SMART approach better than other treatment approaches for prevention of asthma exacerbations? A meta-analysis. Monaldi Arch Chest Dis 2009; 71: 161-9. [Google Scholar] |
| 66. | Cates CJ, Karner C. Combination formoterol and budesonide as maintenance and reliever therapy versus current best practice (including inhaled steroid maintenance), for chronic asthma in adults and children. Cochrane Database Syst Rev 2013; 4: CD007313. [Google Scholar] |
| 67. | Kew KM, Karner C, Mindus SM, Ferrara G. Combination formoterol and budesonide as maintenance and reliever therapy versus combination inhaler maintenance for chronic asthma in adults and children. Cochrane Database Syst Rev 2013; 12: CD009019. [Google Scholar] |
| 68. | Vignola AM. Effects of inhaled corticosteroids, leukotriene receptor antagonists, or both, plus long-acting beta2-agonists on asthma pathophysiology: A review of the evidence. Drugs 2003; 63 Suppl 2:35-51. [Google Scholar] |
| 69. | Bel EH. Clinical Practice. Mild asthma. N Engl J Med 2013; 369: 549-57. [Google Scholar] |
| 70. | Zhang HP, Jia CE, Lv Y, Gibson PG, Wang G. Montelukast for prevention and treatment of asthma exacerbations in adults: Systematic review and meta-analysis. Allergy Asthma Proc 2014; 35: 278-87. [Google Scholar] |
| 71. | Price D, Musgrave SD, Shepstone L, Hillyer EV, Sims EJ, Gilbert RF, et al. Leukotriene antagonists as first-line or add-on asthma-controller therapy. N Engl J Med 2011; 364: 1695-707. [Google Scholar] |
| 72. | Dupont L, Potvin E, Korn D, Lachman A, Dramaix M, Gusman J, et al. Improving asthma control in patients suboptimally controlled on inhaled steroids and long-acting beta2-agonists: Addition of montelukast in an open-label pilot study. Curr Med Res Opin 2005; 21: 863-9. [Google Scholar] |
| 73. | Korn D, Van den Brande P, Potvin E, Dramaix M, Herbots E, Peche R. Efficacy of add-on montelukast in patients with non-controlled asthma: A Belgian open-label study. Curr Med Res Opin 2009; 25: 489-97. [Google Scholar] |
| 74. | Virchow JC, Bachert C. Efficacy and safety of montelukast in adults with asthma and allergic rhinitis. Respir Med 2006; 100: 1952-9. [Google Scholar] |
| 75. | Virchow JC, Mehta A, Ljungblad L, Mitfessel H; MONICA study group. Add-on montelukast in inadequately controlled asthma patients in a 6-month open-label study: The MONtelukast In Chronic Asthma (MONICA) study. Respir Med 2010; 104: 644-51. [Google Scholar] |
| 76. | Ducharme F, Schwartz Z, Hicks G, Kakuma R. Addition of anti-leukotriene agents to inhaled corticosteroids for chronic asthma. Cochrane Database Syst Rev 2004; (2): CD003133. [Google Scholar] |
| 77. | Ducharme FM, Lasserson TJ, Cates CJ. Addition to inhaled corticosteroids of long-acting beta2-agonists versus anti-leukotrienes for chronic asthma. Cochrane Database Syst Rev 2011; (5): CD003137. [Google Scholar] |
| 78. | Guyer AC, Long AA. Long-acting anticholinergics in the treatment of asthma. Curr Opin Allergy Clin Immunol 2013; 13: 392-8. [Google Scholar] |
| 79. | Peters SP, Kunselman SJ, Icitovic N, Moore WC, Pascual R, Ameredes BT, et al. National Heart, Lung, and Blood Institute Asthma Clinical Research Network. Tiotropium bromide step-up therapy for adults with uncontrolled asthma. N Engl J Med 2010; 363: 1715-26. [Google Scholar] |
| 80. | Bateman ED, Kornmann O, Schmidt P, Pivovarova A, Engel M, Fabbri LM. Tiotropium is noninferior to salmeterol in maintaining improved lung function in B16-Arg/Arg patients with asthma. J Allergy Clin Immunol 2011; 128: 315-22. [Google Scholar] |
| 81. | Anderson DE, Kew KM, Boyter AC. Long-acting muscarinic antagonists (LAMA) added to inhaled corticosteroids (ICS) versus the same dose of ICS alone for adults with asthma. Cochrane Database Syst Rev 2015; (8): CD011397. [Google Scholar] |
| 82. | Kerstjens HA, Disse B, Schroder-Babo W, Bantje TA, Gahlemann M, Sigmund R, et al. Tiotropium improves lung function in patients with severe uncontrolled asthma: A randomized controlled trial. J Allergy Clin Immunol 2011; 128: 308-14. [Google Scholar] |
| 83. | Kerstjens HA, Engel M, Dahl R, Paggiaro P, Beck E, Vandewalker M, et al. Tiotropium in asthma poorly controlled with standard combination therapy. N Engl J Med 2012; 367: 1198-207. [Google Scholar] |
| 84. | Tian JW, Chen JW, Chen R, Chen X. Tiotropium versus placebo for inadequately controlled asthma: A meta-analysis. Respir Care 2014; 59: 654-66. doi: 10.4187/respcare.02703. [Google Scholar] |
| 85. | Barnes PJ. Theophylline. Am J Respir Crit Care Med 2013; 188: 901-6. [Google Scholar] |
| 86. | Tee AK, Koh MS, Gibson PG, Lasserson TJ, Wilson AJ, Irving LB. Long acting beta 2-agonists versus theophylline for maintenance treatment of asthma. Cochrane Database Syst Rev 2007; (3): CD001281. [Google Scholar] |
| 87. | Davies B, Brooks G, Devoy M. The efficacy and safety of salmeterol compared to theophylline: Meta-analysis of nine controlled studies. Respir Med 1998; 92: 256-63. [Google Scholar] |
| 88. | Dempsey OJ, Fowler SJ, Wilson A, Kennedy G, Lipworth BJ. Effects of adding either a leukotriene receptor antagonist or low-dose theophylline to a low or medium dose of inhaled corticosteroid in patients with persistent asthma. Chest 2002; 122: 151-9. [Google Scholar] |
| 89. | Evans DJ, Taylor DA, Zetterstrom O, Chung KF, O′Connor BJ, Barnes PJ. A comparison of low-dose inhaled budesonide plus theophylline and high-dose inhaled budesonide for moderate asthma. N Engl J Med 1997; 337: 1412-18. [Google Scholar] |
| 90. | Lim S, Jatakanon A, Gordon D, Macdonald C, Chung KF, Barnes PJ. Comparison of high dose inhaled steroids, low dose inhaled steroids plus low dose theophylline, and low dose inhaled steroids alone in chronic asthma in general practice. Thorax 2000; 55: 837-41. [Google Scholar] |
| 91. | Ukena D, Harnest U, Sakalauskas R, Magyar P, Vetter N, Steffen H, et al. Comparison of addition of theophylline to inhaled steroid with doubling of the dose of inhaled steroid in asthma. Eur Respir J 1997; 10: 2754-60. [Google Scholar] |
| 92. | American Lung Association Asthma Clinical Research Centers. Clinical trial of low-dose theophylline and montelukast in patients with poorly controlled asthma. Am J Respir Crit Care Med 2007; 175: 235-42. [Google Scholar] |
| 93. | Nie H, Zhang G, Liu M, Ding X, Huang Y, Hu S. Efficacy of theophylline plus salmeterol/fluticasone propionate combination therapy in patients with asthma. Respir Med 2013; 107: 347-54. [Google Scholar] |
| 94. | Shukla D, Chakraborty S, Singh S, Mishra B. Doxofylline: A promising methylxanthine derivative for the treatment of asthma and chronic obstructive pulmonary disease. Expert Opin Pharmacother 2009; 10: 2343-56. [Google Scholar] |
| 95. | Goldstein MF, Chervinsky P. Efficacy and safety of doxofylline compared to theophylline in chronic reversible asthma-A double-blind randomized placebo-controlled multicentre clinical trial. Med Sci Monit 2002; 8: CR297-304. [Google Scholar] |
| 96. | Melillo G, Balzano G, Jodice F, De Felice A, Campisi V, Capone M, et al. Treatment of reversible chronic airways obstruction with doxofylline compared with slow-release theophylline: A double-blind, randomized, multicentre trial. Int J Clin Pharmacol Res 1989; 9: 397-405. [Google Scholar] |
| 97. | Normansell R, Walker S, Milan SJ, Walters EH, Nair P. Omalizumab for asthma in adults and children. Cochrane Database Syst Rev 2014; (1): CD003559. [Google Scholar] |
| 98. | Rodrigo GJ, Neffen H, Castro-Rodriguez JA. Efficacy and safety of subcutaneous omalizumab vs placebo as add-on therapy to corticosteroids for children and adults with asthma: A systematic review. Chest 2011; 139: 28-35. [Google Scholar] |
| 99. | Bousquet J, Cabrera P, Berkman N, Buhl R, Holgate S, Wenzel S, et al. The effect of treatment with omalizumab, an anti-IgE antibody, on asthma exacerbations and emergency medical visits in patients with severe persistent asthma. Allergy 2005; 60: 302-8. [Google Scholar] |
| 100. | Holgate ST, Chuchalin AG, Hébert J, Lötvall J, Persson GB, Chung KF, et al. Omalizumab 011 International Study Group. Efficacy and safety of a recombinant anti-immunoglobulin E antibody (omalizumab) in severe allergic asthma. Clin Exp Allergy 2004; 34: 632-8. [Google Scholar] |
| 101. | Kuprys-Lipinska I, Kuna P. Loss of asthma control after cessation of omalizumab treatment: real life data. Postepy Dermatol Alergol 2014; 31: 1-5. [Google Scholar] |
| 102. | Chorao P, Pereira AM, Fonseca JA. Inhaler devices in asthma and COPD-an assessment of inhaler technique and patient preferences. Respir Med 2014; 108: 968-75. [Google Scholar] |
| 103. | Arora P, Kumar L, Vohra V, Sarin R, Jaiswal A, Puri MM, et al. Evaluating the technique of using inhalation device in COPD and bronchial asthma patients. Respir Med 2014;1 08: 992-8. [Google Scholar] |
| 104. | Cates CJ, Welsh EJ, Rowe BH. Holding chambers (spacers) versus nebulisers for beta-agonist treatment of acute asthma. Cochrane Database Syst Rev 2013; (9): CD000052. [Google Scholar] |
| 105. | Bateman ED, Clark TJ, Frith L, Bousquet J, Busse WW, Pedersen SE, et al. Rate of response of individual asthma control measures varies and may overestimate asthma control: An analysis of the goal study. J Asthma 2007; 44: 667-73. [Google Scholar] |
| 106. | Rogers L, Reibman J. Stepping down asthma treatment: How and when. Curr Opin Pulm Med 2012; 18: 70-5. [Google Scholar] |
| 107. | Bacharier LB. Step-down therapy in asthma: A focus on treatment options for patients receiving inhaled corticosteroids and long-acting beta-agonist combination therapy. Allergy Asthma Proc 2012; 33: 13-18. [Google Scholar] |
| 108. | Papi A, Nicolini G, Crimi N, Fabbri L, Olivieri D, Rossi A, et al. Step-down from high dose fixed combination therapy in asthma patients: A randomized controlled trial. Respir Res 2012; 13: 54. [Google Scholar] |
| 109. | Hawkins G, McMahon AD, Twaddle S, Wood SF, Ford I, Thomson NC. Stepping down inhaled corticosteroids in asthma: Randomised controlled trial. BMJ 2003; 326: 1115. [Google Scholar] |
| 110. | Hagan JB, Samant SA, Volcheck GW, Li JT, Hagan CR, Erwin PJ, et al. The risk of asthma exacerbation after reducing inhaled corticosteroids: A systematic review and meta-analysis of randomized controlled trials. Allergy 2014; 69: 510-16. [Google Scholar] |
| 111. | Bateman ED, Jacques L, Goldfrad C, Atienza T, Mihaescu T, Duggan M. Asthma control can be maintained when fluticasone propionate/salmeterol in a single inhaler is stepped down. J Allergy Clin Immunol 2006; 117: 563-70. [Google Scholar] |
| 112. | Godard P, Greillier P, Pigearias B, Nachbaur G, Desfougeres JL, Attali V. Maintaining asthma control in persistent asthma: comparison of three strategies in a 6-month double-blind randomised study. Respir Med 2008; 102: 1124-31. [Google Scholar] |
| 113. | Reddel HK, Gibson PG, Peters MJ, Wark PA, Sand IB, Hoyos CM, et al. Down-titration from high-dose combination therapy in asthma: Removal of long-acting beta(2)-agonist. Respir Med 2010; 104: 1110-20. [Google Scholar] |
| 114. | Koenig SM, Ostrom N, Pearlman D, Waitkus-Edwards K, Yancey S, Prillaman BA, et al. Deterioration in asthma control when subjects receiving fluticasone propionate/salmeterol 100/50 mcg Diskus are ′stepped-down′. J Asthma 2008; 45: 681-7. [Google Scholar] |
| 115. | Berger WE, Bleecker ER, O′Dowd L, Miller CJ, Mezzanotte W. Efficacy and safety of budesonide/formoterol pressurized metered-dose inhaler: Randomized controlled trial comparing once- and twice-daily dosing in patients with asthma. Allergy Asthma Proc 2010; 31: 49-59. [Google Scholar] |
| 116. | Brozek JL, Kraft M, Krishnan JA, Cloutier MM, Lazarus SC, Li JT, et al. Long-acting â2-agonist step-off in patients with controlled asthma. Arch Intern Med 2012; 172: 1365-75. [Google Scholar] |
| 117. | Ahmad S, Kew KM, Normansell R. Stopping long-acting beta2-agonists (LABA) for adults with asthma well controlled by LABA and inhaled corticosteroids. Cochrane Database Syst Rev 2015; (6): CD011306. [Google Scholar] |
| 118. | Masoli M, Weatherall M, Holt S, Beasley R. Budesonide once versus twice-daily administration: Meta-analysis. Respirology 2004; 9: 528-34. [Google Scholar] |
| 119. | Rank MA, Hagan JB, Park MA, Podjasek JC, Samant SA, Volcheck GW, et al. The risk of asthma exacerbation after stopping low-dose inhaled corticosteroids: A systematic review and meta-analysis of randomized controlled trials. J Allergy Clin Immunol 2013; 131: 724-9. [Google Scholar] |
| 120. | Gionfriddo MR, Hagan JB, Hagan CR, Volcheck GW, Castaneda-Guarderas A, Rank MA. Stepping down inhaled corticosteroids from scheduled to as needed in stable asthma: Systematic review and meta-analysis. Allergy Asthma Proc 2015; 36: 262-7. [Google Scholar] |
Fulltext Views
2,128
PDF downloads
287




