Translate this page into:
Successful extracorporeal life support in respiratory failure after copper sulphate ingestion
Corresponding Author:
Jeong Mi Moon
42 Jebong-ro, Dong-gu
South Korea
jmmoon@chonnam.ac.kr
| How to cite this article: CHO YS, Moon JM, Jeong YH, Lee DH, Chun BJ. Successful extracorporeal life support in respiratory failure after copper sulphate ingestion. Natl Med J India 2018;31:83-85 |
Abstract
A 44-year-old woman intentionally ingested a solution of copper sulphate. She had minimal intravascular haemolysis and methemoglobinaemia but developed acute respiratory distress syndrome (ARDS) 4 hours after acute copper sulphate poisoning. This required extracorporeal membrane oxygenation (ECMO) management in the intensive care unit. Subsequently, she improved clinically and was successfully weaned from ECMO. Acute copper sulphate poisoning can cause severe pulmonary toxicity even in the absence of other serious symptoms. Therefore, a physician treating acute copper sulphate poisoning should look out for respiratory symptoms even in the absence of other common symptoms. We suggest early initiation of venovenous ECMO in those with ARDS following copper sulphate poisoning.
Introduction
Copper is one of the most frequently reported metals to which patients are exposed. Copper sulphate, one of the most abundant salts of copper, is used in various products such as fungicides, algaecides, herbicides and insecticides.[1] The marine blue colour of hydrated copper sulphate crystals is a cause of unintentional poisoning because it is attractive to children. However, copper sulphate poisoning in adults can result from intentional self- harm.[2] The ingestion of large amounts of copper commonly causes intravascular haemolysis, haemolytic anaemia, methemo- globinaemia, acute tubular necrosis, hepatotoxicity and rhabdomyolysis, which can ultimately lead to death. However, few previous reports have described pulmonary toxicity-related copper poisoning.
We describe a patient with copper sulphate poisoning who developed acute respiratory distress syndrome (ARDS) as a major clinical feature.
The Case
A 44-year-old female teacher was referred from a primary care hospital to our emergency department approximately 5 hours after intentional ingestion of over 100 g of copper sulphate including as a solution in her science laboratory.
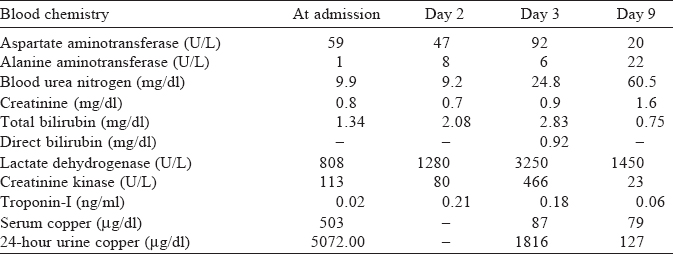
At presentation, her heart rate was 104 per minute, blood pressure was 110/70 mmHg, respiratory rate 26 per minute and her body temperature was 36 °C. She complained of dyspnoea, epigastric pain, nausea and intractable vomiting. Chest X-ray showed diffuse bilateral infiltration [Figure - 1]. The echocardiogram and electro-cardiogram were normal. Her arterial blood gas showed pH 7.23, pCO2 29.7 mmHg, PaO2 68.8 mmHg, HCO3- 12.4 mEq/ L and O2 saturation (SaO2) of 88.9% (on a 10 L non-rebreather face mask). Her initial methemoglobin levels at the time of presentation and 14 hours post-presentation were 1.7% and 5.7%, respectively. The serum copper levels and 24-hour urine copper tests (inductively coupled plasma mass spectrometry) and other biochemical investigations are shown in [Table - 1].
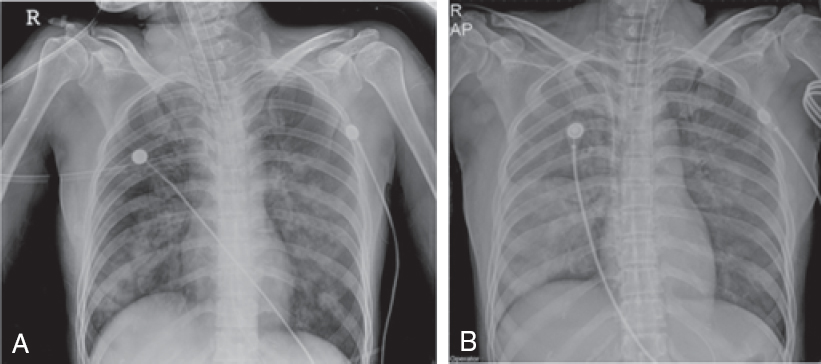 |
| Figure 1: Chest X-rays at the time of admission (A) and after 4 hours (B) |
She received mechanical ventilatory support but her arterial blood gas showed progressive deterioration. After 4 hours, her arterial blood gas showed pH 7.23, pCO2 31.6 mmHg, PaO2 56.2 mmHg, HCO3- 12.9 mEq/L, SaO2 80.8% (with FiO2 1.0) and haemoglobin of 9.9 g/dl. Chest X-ray also showed increased patchy/nodular opacities in both lungs (Fig. 1). She was administered venovenous extracorporeal membrane oxygenation (ECMO) at a flow rate of 3.5 L/minute and a sweep rate of 2 L/ minute. Concurrent ventilator settings included a tidal volume of 350 ml (5 ml/kg), a respiratory rate of 12 breaths/minute, a PEEP of 10 mmHg and an FiO2 of 0.5. Chelation therapy was initiated with penicillamine at a dose of 250 mg every 6 hours because of severe poisoning with more than 100 g of copper sulphate.
Her lung function recovered slowly and the radiological findings also improved by day 7. She could then be weaned from venovenous ECMO and extubated. She was discharged with good clinical recovery.
Discussion
Copper sulphate is a powerful oxidizing agent that exerts corrosive effects on mucous membranes. Ingestion of more than 1 g of copper sulphate can result in clinical manifestations of toxic symptoms.[3] Although the toxicity varies depending on individual factors, the lethal dose is 10-20 g,[2] and mortality is high, reaching up to 25%.[4]·[5] Severe vomiting prevents the absorption of the ingested toxicant, which may partially account for the good outcome of our patient despite the ingestion of 100 g of copper sulphate. The most common features of copper sulphate intoxication are gastrointestinal bleeding and irritation (37%), followed by jaundice (58%), intravascular haemolysis (47%- 65%), acute renal failure (20%-40%), methemoglobinaemia (3.4[0]/0-42[0]/ω) and hepatic encephalopathy (5%).[6]
Our patient was diagnosed to have ARDS,[7] which is defined as the development of acute, bilateral pulmonary infiltrates (as determined by consensus of two trained physician reviewers) and hypoxaemia (PaO2/FIO2 <300 mmHg) not primarily due to heart failure or volume overload. Although respiratory alkalosis should occur primarily in ARDS, this patient showed metabolic acidosis. This may be due to differences in the ARDS phenotype. Recent studies on phenotypes of ARDS have shown that phenotype 2 is characterized by metabolic acidosis, shock, sepsis and higher mortality.[8]·[9]
In copper sulphate poisoning, pulmonary toxicity can occur occasionally and represents an extra-erythrocytic manifestation of the oxidative effects of copper ions.[10] The mechanism underlying pulmonary toxicity induced by copper consists of oxidative effects resulting from the generation of reactive oxygen species (ROS). In addition, copper blocks antioxidant activities in epithelial cells by inhibiting the activity of catalysts and glutathione reductases and by increasing the activity of glutathione peroxidase.[11] Since copper has a high evaporation point (1084 °C), it is difficult to inhale copper at room temperature. In our patient, ARDS might have been caused by aspiration of copper sulphate solution during vomiting.
Few studies have reported on pulmonary toxicity caused by copper poisoning. Giudice et al.[12] reported on dogs exposed to pesticides containing copper sulphate. Acute respiratory failure occurred 3 hours after ingestion, and pulmonary haemorrhage and pneumonia were confirmed by chest X-ray. Pulmonary toxicity was possibly caused by inhalation of copper sulphate. Kim et al.[13] reported a 75-year-old man with vomiting after ingestion of a pesticide containing copper sulphate for suicide. The plasma copper concentration was high at 634.7 mg/ml. The patient’s PaO2 showed hypoxaemia of 60.7 mmHg (room air), and he died of respiratory failure 3 hours after admission. His chest X-ray was normal, and his arterial blood gas showed PaCO2 of 85.2 mmHg and PaO2 of 76.8 mmHg (on 4 L of O2 given through nasal prongs). Although not consistent with the definition of ARDS, pulmonary toxicity can be estimated from hypoxaemia. This patient also had vomiting and most likely aspirated. However, because death occurred 3 hours after presentation, successful therapeutic strategies were not described. In addition, during chronic occupational exposure to copper, interstitial pulmonary fibrosis and histiocytic granulomas that contain copper have been reported.[14] However, pulmonary toxicity related to acute copper poisoning is no longer reported.
In patients with severe ARDS, venovenous ECMO is recommended. The Extracorporeal Life Support Organization (ELSO) recommends ECMO for patients with a risk of death >80%. For patients with FiO2 >90%, Murray scores 2-3 and PaO2/ FiO2 ratio <150, ECMO should be considered.[15],[16] In our patient, ECMO was used with a PaO2/FiO2 ratio of 56 (FiO2 1.0) despite the use of a protective ventilation strategy. For optimal oxygenation in venovenous ECMO, the pump blood flow should be >60% of the theoretical cardiac output, the sweep gas rate should give a PaCO2 between 30 and 40 mmHg, and the oxygen fraction delivered by the extracorporeal circuit (FECO2) should give an arterial oxygen saturation SaO2>88%.[15],[16] Our patient was stabilized at a flow rate of 3.5 L/minute and a sweep rate of 2 L/minute. The ventilator was maintained in lung resting mode.
However, we cannot explain the absence of common symptoms of copper poisoning, such as haemolysis and methemoglobinaemia in our patient. Although the patient’s indirect bilirubin and lactate dehydrogenase increased for 2-3 days, this increase may be explained by the effects of ECMO rather than intravascular haemolysis due to copper poisoning. Intravascular haemolysis has been reported to occur 12-24 hours post-ingestion.[6] Haemolysis is one of the common complications of ECMO and has been reported to occur in 18% of cases.[8]
Supportive treatment is administered for acute copper sulphate intoxication, including the management of corrosive burns, chelation therapy (D-penicillamine, BAL [British anti lewisite], EDTA [calcium disodium ethylene diamine tetra acetic acid] and dimercaprol) and haemodialy sis in those with acute renal failure.[17] Chelation therapy should be initiated when a patient presents with hepatic or haematological complications or other manifestations of poisoning.[10] We started chelation therapy because our patient had ingested more than the reported lethal dose, and ARDS was considered to be a symptom of severe copper poisoning.
Measurements of serum copper concentrations enable a diagnosis of poisoning; however, there is no linear correlation between these values and the clinical features[18] (such as C reactive protein, chest X-ray findings and the PaO2/FiO2 ratio), which can be explained in part by the rapid uptake of copper into red blood cells. In contrast to the development of intravascular haemolysis and hepatotoxicity and the description of renal failure in another report of a patient with a serum copper level of 210 mg/dl, our patient with a level of 503 mg/dl did not have any of these symptoms.[5]
In conclusion, acute copper sulphate poisoning can cause pulmonary toxicity without the well-established symptoms of copper poisoning such as intravascular haemolysis, hepatotoxicity and renal failure. Therefore, a physician treating acute copper poisoning should pay attention to the development of ARDS even in the absence of other common symptoms. In such cases, early initiation ofECMO should be considered for managing respiratory failure.
| 1. | Oldenquist G, Salem M. Parenteral copper sulphate poisoning causing acute renal failure. Nephrol Dial Transplant 1999;14:441–3. [Google Scholar] |
| 2. | Gamakaranage CS, Rodrigo C, Weerasinghe S, Gnanathasan A, Puvanaraj V, Fernando H. Complications and management of acute copper sulphate poisoning; a case discussion. J OccupMed Toxicol 2011;6:34. [Google Scholar] |
| 3. | Sinkovic A, Strdin A, Svensek F. Severe acute copper sulphate poisoning: A case report. Arh Hig Rada Toksikol 2008;59:31-5. [Google Scholar] |
| 4. | Ahasan HA, Chowdhury MA, Azhar MA, Rafiqueuddin AK. Copper sulphate poisoning. Trop Doct 1994;24:52–3. [Google Scholar] |
| 5. | Kayacan SM, Vatansever S, Akkaya V, Erk O, Tukek T, Kýlýcaslan I, et al. A case of copper sulphate intoxication that is presented with prolonged hemolysis and acute renal failure. J Istanbul Faculty Med 2007;70:47–50. [Google Scholar] |
| 6. | Saravu K, Jose J, Bhat MN, Jimmy B, Shastry B. Acute ingestion of copper sulphate: A review on its clinical manifestations and management. Indian J Crit Care Med 2007;11:74. [Google Scholar] |
| 7. | Ranieri VM, Rubenfeld GD, Thompson BT, Ferguson ND, Caldwell E, Fan E, et al. Acute respiratory distress syndrome: The Berlin definition. JAMA 2012;307: 2526-33. [Google Scholar] |
| 8. | Zangrillo A, Landoni G, Biondi-Zoccai G, Greco M, Greco T, Frati G, et al. A meta- analysis of complications and mortality of extracorporeal membrane oxygenation. Crit Care Resusc 2013;15:172–8. [Google Scholar] |
| 9. | 9 Calfee CS, Delucchi K, Parsons PE, Thompson BT, Ware LB, Matthay MA, et al. Subphenotypes in acute respiratory distress syndrome: Latent class analysis of data from two randomised controlled trials. Lancet Respir Med 2014;2:611-20. [Google Scholar] |
| 10. | Hoffman RS, Howland MA, Lewin NA, Nelson L, Goldfrank LR, Flomenbaum N. Goldfrank ‘s toxicologic emergencies. 10th ed. New York:McGraw-Hill; 2015:1882. [Google Scholar] |
| 11. | Rahman I, Biswas SK, Kode A. Oxidant and antioxidant balance in the airways and airway diseases. Eur J Pharmacol 2006;533:222-39. [Google Scholar] |
| 12. | Giudice E, Crinò C, Lanzafame P, Vigano F, Macrì F, Di Pietro S. Acute hypoxemic respiratory failure with hemoptysis in a dog exposed to copper sulphate powder. Top Companion Anim Med 2017;32:36^-0. [Google Scholar] |
| 13. | Kim GB. Acute respiratory failure due to fatal acute copper sulphate poisoning: A case report. J Korean Soc Clin Toxicol 2015;13:36-9. [Google Scholar] |
| 14. | Pimentel JC, Marques F. ‘Vineyard sprayer’s lung’: A new occupational disease. Thorax 1969;24:678-88. [Google Scholar] |
| 15. | Sen A, Callisen H, Alwardt C, Larson J, Lowell A, Libricz S, et al. Adult venovenous extracorporeal membrane oxygenation for severe respiratory failure: Current status and future perspectives. Ann CardAnaesth 2016;19:97–111. [Google Scholar] |
| 16. | Richard C, Argaud L, Blet A, Boulain T, Contentin L, Dechartres A, et al. Extracorporeal life support for patients with acute respiratory distress syndrome: Report of a consensus conference. Ann Intensive Care 2014;4:15. [Google Scholar] |
| 17. | Faure A, Mathon L, Poupelin J, Allaouchiche B, Chassard D. [Acute cupric sulphate intoxication: Pathophysiology and therapy about a case report]. Ann Fr Anesth Reanim 2003;22:557-9. [Google Scholar] |
| 18. | Gunay N, Yildirim C, Karcioglu O, Gunay NE, Yilmaz M, Usalan C, et al. A series of patients in the emergency department diagnosed with copper poisoning: Recognition equals treatment. Tohoku J Exp Med 2006;209:243–8. [Google Scholar] |
Fulltext Views
1,819
PDF downloads
1,966




