Translate this page into:
Syndromic management of sexually transmitted infections: A critical appraisal and the road ahead
Corresponding Author:
Vinod Kumar Sharma
Department of Dermatology and Venereology, All India Institute of Medical Sciences, Ansari Nagar, New Delhi 110029
India
aiimsvks@yahoo.com
| How to cite this article: Gupta V, Sharma VK. Syndromic management of sexually transmitted infections: A critical appraisal and the road ahead. Natl Med J India 2019;32:147-152 |
Abstract
The syndromic approach has been the cornerstone of management of sexually transmitted infections (STIs) in developing countries. This strategy has had a considerable impact in decreasing the burden of STI in society. It offers the advantages of treating the infection at the first visit itself, reducing the risk of complications, non-reliance on laboratory diagnostics, and easy integration into the primary healthcare system. Nonetheless, it is not without limitations, most often criticized for its inability to treat asymptomatic cases. Syndromic management has been found to be satisfactory for genital ulcer disease and urethral discharge in several settings. However, its performance is not as good in the treatment of vaginal discharge syndrome, as it does not allow a distinction between cervicitis and vaginitis. Diagnostic validation and a review of its performance should be done periodically to keep abreast of the changing aetiology of various syndromes and patterns of drug susceptibility. Supplementing the syndromic approach with point-of-care tests and simple laboratory tests where available can improve its results. Further, healthcare professionals should be imparted training for optimum patient care. This narrative review critically appraises the syndromic approach to STIs, discusses the challenges that it faces, and offers suggestions to improve its performance.
Introduction
Sexually transmitted infections (STIs) continue to be a major public health problem, especially in the post-HIV era. About 357 million new cases of curable STIs (gonorrhoea, chlamydia, syphilis and trichomoniasis) occur in adults every year. More than 90% of STIs occur in low-, lower-middle- and upper-middle income countries.[1] In India, a community-based prevalence study conducted by the Indian Council of Medical Research reported that over 6% of the adult population suffers from an episode of STI or reproductive tract infection (RTI) every year. This would translate into about 33 million STI episodes annually.[2]
It is known that several STIs increase the risk of transmission of HIV, and studies have shown that a better control of STIs can prevent the spread of HIV.[3],[4] Prompt diagnosis and treatment is essential for the control and prevention of STIs. Conventionally, there are two approaches to the diagnosis of STI: aetiological or clinical diagnosis. The most accurate way of diagnosing STI, or any infection for that matter, relies on the use of laboratory-based diagnostic techniques to identify the causative organism. However, this approach is expensive (requires trained laboratory personnel, equipment, storage and transport facilities) and leads to delays in diagnosis and treatment. Moreover, these diagnostic tests are not widely available in resource-poor settings where STIs are a major health burden. On the other hand, diagnosis of an STI based solely on the clinical presentation is unreliable.[5],[6] Considering the limitations of laboratory-based aetiological as well as clinical diagnosis, the concept of a ‘syndromic approach’ was introduced, and syndromic management guidelines were promoted by WHO in 1991 to tackle the STI epidemic.
What is the Syndromic Approach?
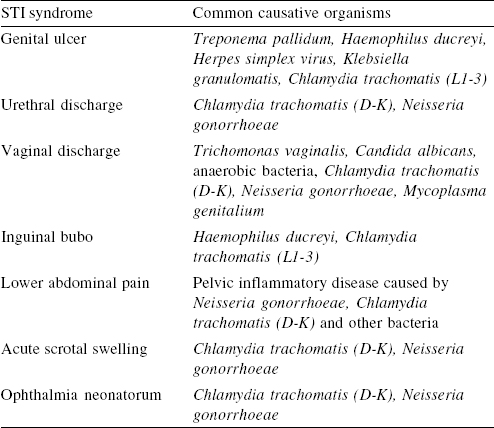
Although STIs are caused by a variety of microorganisms, the signs and symptoms related to STIs can be grouped into a limited number of ‘syndromes’. A syndrome is a set of clinically distinct signs and symptoms that can be easily recognized by the clinician. Syndromic diagnosis refers to identification of a consistent group of symptoms and easily recognized signs [Table - 1].[7] Syndromic treatment refers to treating common pathogens for a particular syndrome. For example, the common causative organisms for urethral discharge syndrome are Chlamydia trachomatis and Neisseria gonorrhoeae, and therefore, syndromic treatment should cover both these pathogens.
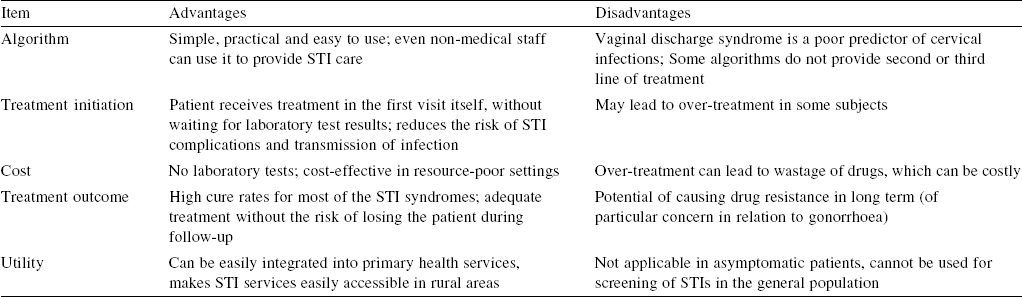
Syndromic management of STIs includes the identification of the syndrome on the basis of the patient’s symptoms and signs and using the available flowcharts to treat it. The syndromic flowcharts are simple and easy to use for healthcare providers and, once trained, even non-STI specialists can use them at primary health centres in rural or remote areas. This approach also includes contact tracing and partner management and a return visit to ensure treatment compliance. It provides an opportunity to educate and counsel patients regarding safe sex practices. All patients should be referred for voluntary counselling and testing for HIV, syphilis and hepatitis B.[8] The main components of syndromic management of STIs are summarized in [Table - 2].[9] Even mixed infections get treated with this approach, and spread of infection is curtailed by reducing the duration of illness. As syndromic management does not wait for results of laboratory tests, it leads to immediate treatment. However, simple laboratory tests, wherever available, should also be done as they can increase the sensitivity, specificity and predictive values of this ‘enhanced’ syndromic approach. The advantages and disadvantages of the syndromic approach to STIs are summarized in [Table - 3].[7]

The syndromic flowcharts for STIs are based on the prevalence of STIs, common pathogens, and drug availability and susceptibility patterns in a particular area. Many countries have developed their own flowcharts, whereas WHO-recommended flowcharts can be used in countries that do not have their own treatment algorithm.
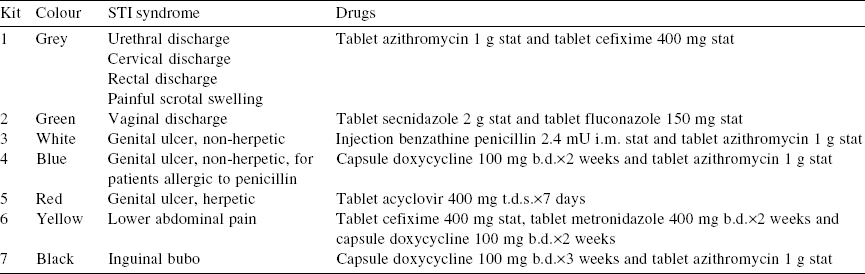
In India, the flowcharts recommended by the National AIDS Control Organization (NACO) are available for use at primary health centres as well as designated STI/RTI clinics (Suraksha clinics) at district hospitals and medical college hospitals. There are 1160 such clinics (situated at government health facilities at the district level and above) throughout the country. Under the National AIDS Control Programme (NACP)-IV, 10 regional STI laboratories and 45 state reference laboratories are tasked with validation of the syndromic approach. The NACO provides prepacked colour-coded treatment kits under the NACP for syndromic management of STIs [Table - 4]. These treatment kits have been developed on the basis of the National Guidelines on Prevention, Management and Control of Reproductive Tract Infections including STIs (issued by the Ministry of Health and Family Welfare), and are available free of cost at various public facilities offering STI services.
Effectiveness of the Syndromic Approach
One of the first evidences in favour of syndromic management was perhaps provided by the Mwanza Intervention Trial in 1997. The prevalence of serological syphilis and symptomatic urethritis in communities receiving STI care based on the syndromic approach decreased by 30% and 50%, respectively.[10] Further, there was a 42% reduction in the new HIV cases in this group compared to the control group.[3] Since then, the operational performance of the syndromic approach and its utility in the diagnosis, treatment and prevention of various STIs have been evaluated in various settings. The syndromic approach has been effective in decreasing the burden of STIs.[11] The rates of STI have reduced considerably in areas where control strategies based on the syndromic approach have been implemented, such as in sex workers in Cote d’Ivoire, Senegal and South Africa, and in STI clinics in Kenya and Burkina Faso.[11] In fact, the impact of the syndromic approach goes beyond the treated population; decreasing rates of STIs have been observed even in the general population.[12],[13]
While the syndromic approach in genital ulcer and urethral discharge syndromes has been found to be effective, its performance in vaginal discharge syndrome has been underwhelming. Pettifor et al.[14] reviewed studies on syndromic management of STIs, published till 1998. The sensitivity of the WHO algorithms for syphilis and chancroid was high, ranging from 72% to 100%; however, it was low for genital herpes (4.5%) in a study from Rwanda. The overall cure rates were also high, ranging from 68% to 100%. A study conducted in eight STI clinics (Delhi, Maharashtra, Andhra Pradesh and Manipur) in India reported the overall sensitivity and specificity of the syndromic algorithm for genital ulcer syndrome (n=194) to be 68% and 52%, respectively, and the positive predictive value 50%. The sensitivity of the algorithm for herpetic genital ulcer disease was better than that of non-herpetic genital ulcer disease (74% v. 33%), whereas specificity was lower (33% v. 56%). Importantly, 52 (42%) cases were misclassified: genital herpes was more commonly misdiagnosed using the syndromic approach (n=34) than syphilis and other infections (n=18).[15] Another study from Gujarat reported high sensitivity and poor specificity of the syndromic approach for herpetic genital ulcer disease. Of the 96 cases with genital ulcer disease, 71 were clinically diagnosed as herpetic and 25 as non-herpetic. Only 46% (n=33/71) of cases of herpetic genital ulcer could be validated. While it did not miss any case of genital herpes, about 54% of cases would have been over-treated for herpes. On the other hand, a majority (88%, n=22/25) of the non- herpetic genital ulcers were diagnosed correctly, but about 34% of cases would have been missed if laboratory tests had not been done.[16]
The syndromic approach seemingly works well for urethral discharge syndrome, with sensitivity ranging from 87% to 99% and cure rates from 92% to 99%. Specificity ranged from 7% to 90%, implying that patients were over-treated for gonococcal and chlamydial infections.[14] A study from Manaus, Brazil, reported an overall cure rate of 98% (n=620/633) in patients with urethral discharge syndrome with syndromic management: 91.5% with first-line algorithm and an additional 6.5% with second-line algorithm.[17] However, the performance of urethral discharge syndrome in a few small studies from India has been variable, with sensitivity ranging from 55% to 88%.[16],[18] The use of microscopy can increase the specificity and positive predictive value of the syndromic approach, while decreasing its sensitivity marginally.[19],[20]
Most of the studies evaluating the performance of vaginal discharge syndrome included in the review by Pettifor et al.[14] have tested its ability to detect cervical infection. The sensitivity ranged from 73% to 93%, and cure rates ranged from 86% to 96%.[14] Addition of speculum examination led to a substantial drop in the sensitivity, while increasing the specificity only marginally.[21],[22] However, it cannot distinguish between vaginal and cervical infections and results in over-treatment for cervical infections. Incorporation of risk assessment scores can reduce over-treatment for cervical infection. This strategy reduced the over-treatment rate from 92% to 17% in pregnant women and from 89% to 36% in non-pregnant women in Tanzania.[23] A specificity of 93% was reported for a syndromic algorithm based on risk assessment scores and speculum-assisted evaluation for cervical infections in a study from Delhi (n=319), but the sensitivity was only 5%. However, it had an excellent sensitivity for the treatment of bacterial vaginosis (94%) and trichomoniasis (100%).[24] Another review of studies published from 2001 onwards reiterated that vaginal discharge syndrome flowcharts work well for only vaginal infections, whereas its performance for cervical infections is poor, with a high proportion of over- as well as missed treatment.[25] In a study in the obstetrics and gynaecology departments of different hospitals in Delhi, only 19% (n=344/1797) of women with vaginal discharge tested positive for an STI organism.[26] Such a disparity between syndromic and aetiological diagnosis had been reported earlier as well, suggesting that the syndromic approach over-diagnoses vaginal discharge as an STI, especially in a low-prevalence setting.[27],[28] Clearly, all vaginal discharges are not STIs. A community-based study from Goa found psychosocial factors to have the strongest association with vaginal discharge, whereas RTI/STIs were not associated with this complaint.[29] Vaginal discharge syndrome performs better in high-risk population compared to the general population because cervical infections are more likely in such a population. A study conducted among female sex workers in Surat, India, reported a sensitivity of vaginal discharge syndrome for cervicitis to be 54%, whereas specificity was 49% and positive predictive value was 21%.[30] On combining treatment for trichomoniasis, the sensitivity and specificity increased marginally to 65% and 55%, respectively, but the positive predictive value almost doubled to 41%. Despite its relatively poor performance, the sensitivity and positive predictive values were better in this high-risk population.[30] A recommendation could be made for treating women with vaginal discharge for vaginal infection only, unless the patient profile suggests risk for cervicitis when treatment for cervical infection should also be given.
Challenges and Future Directions
One of the biggest criticisms of the syndromic approach has been its inability to detect asymptomatic cases. A large proportion of infected individuals are asymptomatic, and missing these asymptomatic infections at the population level results in ongoing transmission, delayed sequelae and long-term complications.[31] A study from South Africa showed that clinical assessment missed about 88% of laboratory-diagnosed STIs in women at the time of HIV diagnosis.[32] Aggressive strategies such as mass treatment have been suggested to overcome this issue. Korenromp et al. used a stochastic simulation model which projected that a single- round mass treatment had a greater short-term impact on HIV compared to the syndromic approach (36% v. 30% at 2 years), but a smaller long-term impact (24% v. 62% at 10 years). On combining mass treatment with syndromic treatment, the authors found a rapid and sustained fall in the incidence of HIV (57% at 2 years, 70% at 10 years).[33] Such an approach would take care of asymptomatic patients; there is no need for contact tracing, and treatment compliance and effectiveness are assured but there is risk of antibiotic resistance.[34]
Digital technology and the internet can also be used to provide low-cost, engaging and deeply permeating STI/HIV prevention practices at the community level.[35] Focus is now shifting towards a diagnostic model of STI care, given the improving economic status of developing countries, availability of affordable point-of- care tests and the possible use of expedited partner therapy (EPT). In EPT, the patient is given the treatment or prescription for his/ her sexual partner(s). This approach can reduce the risk of chlamydial re-infection in women by 20%-29%. It has been used in the UK, USA and Australia, with encouraging results, but requires further systematic evaluation.[36] An STI care model comprising point-of-care testing, immediate treatment and EPT was tested in 267 HIV-negative women in South Africa in 201617. The EPT acceptance rate was 87%, and 89% of these women reported that their partner had taken the medicine. The detection of infection at 6-weeks follow-up in women receiving EPT was much lower than those who did not (2.2% v. 40%, p=0.023).[37]
The syndromic approach needs to be frequently reviewed to ascertain whether it is performing satisfactorily in the community. The success of the syndromic approach depends on several factors such as the epidemiology of the causative microorganisms in a given population, drug susceptibility of the pathogens and background prevalence of STIs and HIV. These are ever-changing variables which require regular monitoring, so that the algorithms can be adjusted accordingly. In South Africa, several key revisions were made to the national STI syndromic flowcharts based on many STI surveys.[38] Because of the increasing resistance to quinolones among N. gonorrhoeae isolates observed in many South African cities, guidelines were changed to cephalosporins for the treatment of presumptive gonorrhoea. Acyclovir was added as part of first-line treatment for genital ulcers, considering the rising prevalence of genital herpes.[38] Such policy changes can be made only when robust national-level data are available. However, the STI reporting system in India suffers from several lacunae. Most of the information is based on syndromic data, reported passively from the designated STI/RTI clinics with limited laboratory facilities catering to symptomatic patients. Only the regional STI training and reference centres provide information on aetiological diagnosis and antimicrobial susceptibility. Further, there are no clear guidelines on reporting patients with STIs who visit health facilities other than the designated STI centres, such as other departments of a government hospital (e.g. gynaecology), private practitioners or alternative systems of medicine. Therefore, information on STI epidemiology in India is largely incomplete and inaccurate, and cannot be used to validate the syndromic approach. In view of these limitations, Haldar et al. proposed a framework for sentinel surveillance in India, building on the existing infrastructure.[39] A district can be made a ‘sentinel unit’ wherein the designated STI clinics as well as private centres would function as sentinel reporting sites. This approach can help provide a greater supervisory support and regular monitoring, thereby ensuring a high level of data quality.[39] Diagnostic validation of the syndromic approach should also be done periodically. Several studies have shown poor agreement of the syndromic approach with laboratory-based diagnostics in various settings, especially for vaginal discharge syndrome.[27],[40],[41],[42] Laboratory testing, when available, should be combined with the syndromic approach as it can improve its specificity and positive predictive value. A study from Kisumu, Kenya, reported poor agreement between syndromic and aetiological diagnoses of STIs (κ=0.09). The syndromic approach missed the diagnosis of STI in a majority of cases: only 10.4% (n=88) of patients were diagnosed as STI on the basis of clinical symptoms compared to 32.2% (n=272) on the basis of laboratory tests. Herpes simplex virus (HSV)-2 infection was severely underdiagnosed, whereas chlamydia and gonorrhoea were over-diagnosed using the syndromic approach.[42]
As the performance of syndromic algorithms depends on the aetiological spectrum of the syndrome, these should be periodically revised and adapted to the changing epidemiological patterns of STI in a given setting. There has been a gradual shift in the aetiology of genital ulcer disease—syphilis and chancroid are no longer the leading causes of sexually transmitted genital ulcers. Several studies around the world have documented HSV as the most common cause of genital ulcer disease in the present HIV era, with its prevalence ranging from 48% in India to 55% in Brazil, 61% in Uganda and 74% in South Africa.[15],[43],[44],[45] A 9-year (2007-15) study from South Africa reported the prevalence of HSV ranging from 48% to 75% in patients with genital ulcer disease.[46] A study from Ethiopia reported failure of the syndromic approach in 30% of women, which was attributed to the high prevalence of HSV infection as treatment algorithms did not include treatment for genital herpes.[47] A study from Botswana reported the sensitivity of the algorithm of genital ulcer syndrome to increase from 33% to 99% following addition of treatment for herpes for all cases of genital ulcer disease. The missed infection rate decreased from 67% to 1%.[48] In 2003, WHO recommended inclusion of treatment for genital herpes in the syndromic flowcharts if HSV accounts for 30% or more of genital ulcer disease,[49] which was subsequently modified in 2008 to include antiherpetic treatment as part of the first-line therapy in all settings without a prevalence threshold.[50] In India, the NACO flowcharts for genital ulcer syndrome are currently classified separately into herpetic and non-herpetic causes. Given that HSV is the most common cause of genital ulcer disease coupled with the fact that it is frequently missed clinically, inclusion of treatment for genital herpes should be considered in the syndromic algorithms for genital ulcer disease in India as well.
There is a need to train healthcare professionals in providing the STI/RTI services, so that the syndromes are not misdiagnosed and correct treatment is offered. A study from Gujarat identified gaps in the knowledge and application of enhanced syndromic management at various levels of healthcare. The paramedical staff (nurses and laboratory technicians) was found to have poor basic knowledge of RTI/STI, signs and symptoms, examination, identification of syndromes, the syndromic approach and HIV- related and other complications. On the other hand, doctors (55% were qualified postgraduates, about 33% represented tertiary- level care) had a satisfactory basic knowledge regarding RTI/STI and identification of syndromes, but it was poor for examination, syndromic management, complications and HIV. The poor baseline knowledge among the paramedical staff was alarming as they are the ones usually involved in providing information and counselling to the patients. Surprisingly, even doctors had poor knowledge of management issues. Doctors and paramedical staff were given training over a period of 2-3 days through lectures, group discussions, case studies and role play, with improvement in some of the deficient areas.[51] Another study from Peru evaluated the effect of training (seminars, workshops and continuing medical education) on syndromic management through simulated patients. This training was given in 10 cities, and the results were compared with 10 control cities at baseline, 3-, 6- and 18-months post training for three STI syndromes (urethral discharge, vaginal discharge and genital ulcer disease). There was no significant difference between the intervention and control cities at baseline, but the performance of intervention cities improved significantly for all measures of management at all follow-up visits.[52] Such training programmes need to be regularly conducted to consolidate these short- to mid-term gains. Suggestions for improving the performance of syndromic case management are listed in [Table - 5].

Is the Syndromic Approach Relevant Today?
Syndromic management has been the cornerstone of STI care where it is most needed—in developing countries. However, studies have suggested poor agreement of syndromic diagnosis with aetiological diagnosis and poor operational efficacy, especially in areas with high HIV-prevalence. Ironically, it is the areas with high HIV-burden where good-quality STI care is needed the most owing to the epidemiological and biological interactions between STIs and HIV. A study from KwaZulu-Natal, at the epicentre of the HIV epidemic, reported an overall median effectiveness of syndromic management in only 13% of symptomatic curable STI episodes between 1987 and 2004.[53] Further, it remains a poor screening approach when applied to asymptomatic cases, particularly in women. Despite these limitations, the syndromic approach remains the most feasible method of STI management in low-income countries, until inexpensive, simple and rapid point- of-care STI diagnostic tests are made available for use in these settings.
Till such time, the following changes in the syndromic approach are needed immediately for it to still remain relevant: (i) a separate strategy to detect and treat asymptomatic cases for overall control of STI burden in the community; (ii) revision of syndromic algorithms as per the current STI trends and drug susceptibility patterns; (iii) widespread use of the ‘enhanced’ syndromic approach wherever possible; and (iv) regular training and monitoring of medical and paramedical staff, with a focus on examination and counselling in addition to providing treatment kits.
Conflicts of interest. None declared
| 1. | Newman L, Rowley J, Vander Hoorn S, Wijesooriya NS, Unemo M, Low N, et al. Global estimates of the prevalence and incidence of four curable sexually transmitted infections in 2012 based on systematic review and global reporting. PLoS One 2015; 10:e0143304. [Google Scholar] |
| 2. | Burden of STI/RTI infection in India. Indian Council of Medical Research. As cited in Annual Report of 2015-16. Ministry of Health and Family Welfare, Government of India; 2002-03. p. 35. [Google Scholar] |
| 3. | Grosskurth H, Mosha F, Todd J, Mwijarubi E, Klokke A, Senkoro K, et al. Impact of improved treatment of sexually transmitted diseases on HIV infection in rural Tanzania: Randomised controlled trial. Lancet 1995;346:530-6. [Google Scholar] |
| 4. | Dallabetta G, Neilson G. Efforts to control sexually transmitted infections as a means to limit HIV transmission: What is the evidence? Curr HIV/AIDS Rep 2004;1: 166-71. [Google Scholar] |
| 5. | Dangor Y, Ballard RC, da L Exposto F, Fehler G, Miller SD, Koornhof HJ. Accuracy of clinical diagnosis of genital ulcer disease. Sex Transm Dis 1990; 17:184-9. [Google Scholar] |
| 6. | Ndinya-Achola JO, Kihara AN, Fisher LD, Krone MR, Plummer FA, Ronald A, et al. Presumptive specific clinical diagnosis of Genital Ulcer Disease (GUD) in a primary health care setting in Nairobi. Int J STD AIDS 1996;7:201-5. [Google Scholar] |
| 7. | Bosu WK. Syndromic management of sexually transmitted diseases: Is it rational or scientific? Trop Med Int Health 1999;4:114-9. [Google Scholar] |
| 8. | World Health Organization. Training modules for the syndromic management of sexually transmitted infections. 2nd ed. Geneva:World Health Organization; 2007. [Google Scholar] |
| 9. | Management of patients with sexually transmitted diseases. Report of a WHO Study Group. World Health Organ Tech Rep Ser 1991;810:1-103. [Google Scholar] |
| 10. | Mayaud P, Mosha F, Todd J, Balira R, Mgara J, West B, et al. Improved treatment services significantly reduce the prevalence of sexually transmitted diseases in rural Tanzania: Results of a randomized controlled trial. AIDS 1997;11:1873-80. [Google Scholar] |
| 11. | Vuylsteke B. Current status of syndromic management of sexually transmitted infections in developing countries. Sex Transm Infect 2004;80:333-4. [Google Scholar] |
| 12. | Honey E, Augood C, Templeton A, Russell I, Paavonen J, Mårdh PA, et al. Cost effectiveness of screening for Chlamydia trachomatis: A review of published studies. Sex Transm Infect 2002;78:406-12. [Google Scholar] |
| 13. | Chief Medical Officer’s Expert Advisory Group. Main Report of the CMO’s Expert Advisory Group on Chlamydia trachomatis. London:Department of Health; 1998. [Google Scholar] |
| 14. | Pettifor A, Walsh J, Wilkins V, Raghunathan P. How effective is syndromic management of STDs? A review of current studies. Sex Transm Dis 2000;27: 371-85. [Google Scholar] |
| 15. | Prabhakar P, Narayanan P, Deshpande GR, Das A, Neilsen G, Mehendale S, et al. Genital ulcer disease in India: Etiologies and performance of current syndrome guidelines. Sex Transm Dis 2012;39:906-10. [Google Scholar] |
| 16. | Bhavsar C, Patel RM, Marfatia Y. A study of 113 cases of genital ulcerative disease and urethral discharge syndrome with validation of syndromic management of sexually transmitted diseases. Indian J Sex Transm Dis AIDS 2014;35:35-9. [Google Scholar] |
| 17. | Menezes Filho JR, Sardinha JC, Galbán E, Saraceni V, Talhari C. Effectiveness of syndromic management for male patients with urethral discharge symptoms in Amazonas, Brazil. An Bras Dermatol 2017;92:779-84. [Google Scholar] |
| 18. | Das A, Ghosh P, Ghosh I, Bhattacharya R, Azad Sardar AK, Goswami S, et al. Usefulness and utility of NACO regime in the management of sexually transmitted infections: A pilot study. Indian J Dermatol 2017;62:630–4. [Google Scholar] |
| 19. | Alary M, Baganizi E, Guèdèmè A, Padonou F, Davo N, Adjovi C, et al. Evaluation of clinical algorithms for the diagnosis of gonococcal and chlamydial infections among men with urethral discharge or dysuria and women with vaginal discharge in Benin. Sex Transm Infect 1998;74 (Suppl 1):S44-9. [Google Scholar] |
| 20. | Moherdaui F, Vuylsteke B, Siqueira LF, dos Santos Júnior MQ, Jardim ML, de Brito AM, et al. Validation of national algorithms for the diagnosis of sexually transmitted diseases in Brazil: Results from a multicentre study. Sex Transm Infect 1998;74 (Suppl 1):S38-43. [Google Scholar] |
| 21. | Diallo MO, Ghys PD, Vuylsteke B, Ettiègne-Traoré V, Gnaoré E, Soroh D, et al. Evaluation of simple diagnostic algorithms for Neisseria gonorrhoeae and Chlamydia trachomatis cervical infections in female sex workers in Abidjan, Côte D’ivoire. Sex Transm Infect 1998;74 (Suppl 1):S106-11. [Google Scholar] |
| 22. | Vuylsteke B, Laga M, Alary M, Gerniers MM, Lebughe JP, Nzila N, et al. Clinical algorithms for the screening of women for gonococcal and chlamydial infection: Evaluation of pregnant women and prostitutes in Zaire. Clin Infect Dis 1993;17: 82-8. [Google Scholar] |
| 23. | Mayaud P, Ka-Gina G, Cornelissen J, Todd J, Kaatano G, West B, et al. Validation of a WHO algorithm with risk assessment for the clinical management of vaginal discharge in Mwanza, Tanzania. Sex Transm Infect 1998;74 (Suppl 1):S77-84. [Google Scholar] |
| 24. | Vishwanath S, Talwar V, Prasad R, Coyaji K, Elias CJ, de Zoysa I. Syndromic management of vaginal discharge among women in a reproductive health clinic in India. Sex Transm Infect 2000;76:303-6. [Google Scholar] |
| 25. | Zemouri C, Wi TE, Kiarie J, Seuc A, Mogasale V, Latif A, et al. The performance of the vaginal discharge syndromic management in treating vaginal and cervical infection: A systematic review and meta-analysis. PLoS One 2016;11:e0163365. [Google Scholar] |
| 26. | Sonkar SC, Wasnik K, Kumar A, Sharma V, Mittal P, Mishra PK, et al. Evaluating the utility of syndromic case management for three sexually transmitted infections in women visiting hospitals in Delhi, India. Sci Rep 2017;7:1465. [Google Scholar] |
| 27. | Chauhan V, Shah M, Thakkar S, Patel SV, Marfatia Y. Sexually transmitted infections in women: A correlation of clinical and laboratory diagnosis in cases of vaginal discharge syndrome. Indian Dermatol Online J 2014;5:S1-5. [Google Scholar] |
| 28. | Ray K, Muralidhar S, Bala M, Kumari M, Salhan S, Gupta SM, et al. Comparative study of syndromic and etiological diagnosis of reproductive tract infections/sexually transmitted infections in women in Delhi. Int J Infect Dis 2009;13:e352-9. [Google Scholar] |
| 29. | Patel V, Pednekar S, Weiss H, Rodrigues M, Barros P, Nayak B, et al. Why do women complain of vaginal discharge? A population survey of infectious and pyschosocial risk factors in a South Asian community. Int J Epidemiol 2005;34: 853-62. [Google Scholar] |
| 30. | Desai VK, Kosambiya JK, Thakor HG, Umrigar DD, Khandwala BR, Bhuyan KK. Prevalence of sexually transmitted infections and performance of STI syndromes against aetiological diagnosis, in female sex workers of red light area in Surat, India. Sex Transm Infect 2003;79:111-15. [Google Scholar] |
| 31. | Moodley D, Moodley P, Sebitloane M, Soowamber D, McNaughton-Reyes HL, Groves AK, et al. High prevalence and incidence of asymptomatic sexually transmitted infections during pregnancy and postdelivery in KwaZulu Natal, South Africa. Sex Transm Dis 2015;42:43-7. [Google Scholar] |
| 32. | Mlisana K, Naicker N, Werner L, Roberts L, van Loggerenberg F, Baxter C, et al. Symptomatic vaginal discharge is a poor predictor of sexually transmitted infections and genital tract inflammation in high-risk women in South Africa. J Infect Dis 2012;206:6-14. [Google Scholar] |
| 33. | Korenromp EL, Van Vliet C, Grosskurth H, Gavyole A, Van der Ploeg CP, Fransen L, et al. Model-based evaluation of single-round mass treatment of sexually transmitted diseases for HIV control in a rural African population. AIDS 2000;14:573-93. [Google Scholar] |
| 34. | Sen S. Syndromic management in the control of sexually transmitted infections: Time for a relook. Indian J Dermatol Venereol Leprol 2013;79:816-17. [Google Scholar] |
| 35. | Swendeman D, Rotheram-Borus MJ. Innovation in sexually transmitted disease and HIV prevention: Internet and mobile phone delivery vehicles for global diffusion. Curr Opin Psychiatry 2010;23:139–44. [Google Scholar] |
| 36. | Ferreira A, Young T, Mathews C, Zunza M, Low N. Ferreira A, Young T, Mathews C, Zunza M, Low N. Strategies for partner notification for sexually transmitted infections, including HIV. Cochrane Database Syst Rev 2013;(10):CD002843. [Google Scholar] |
| 37. | Garrett NJ, Osman F, Maharaj B, Naicker N, Gibbs A, Norman E, et al. Beyond syndromic management: Opportunities for diagnosis-based treatment of sexually transmitted infections in low- and middle-income countries. PLoS One 2018;13:e0196209. [Google Scholar] |
| 38. | Lewis DA, Maruma E. Revision of the national guideline for first-line comprehensive management and control of sexually transmitted infections: What’s new and why? South Afr J Epidemiol Infect 2009;24:6-9. [Google Scholar] |
| 39. | Haldar P, Morineau G, Das A, Mehendale S. A surveillance model for sexually transmitted infections in India. Indian J Public Health 2015;59:286-94. [Google Scholar] |
| 40. | Vallely LM, Toliman P, Ryan C, Rai G, Wapling J, Gabuzzi J, et al. Performance of syndromic management for the detection and treatment of genital Chlamydia trachomatis, Neisseria gonorrhoeae and Trichomonas vaginalis among women attending antenatal, well woman and sexual health clinics in Papua New Guinea: A cross-sectional study. BMJ Open 2017;7:e018630. [Google Scholar] |
| 41. | Arif N, Sebastian S, Khanna N, Sood S, Dhawan B. Comparative analysis of syndromic case management and polymerase chain reaction based diagnostic assays for treatment of Neisseria gonorrhoeae, Chlamydia trachomatis and genital mycoplasmas in patients of genitourinary discharge. Indian J Med Microbiol 2017; 35:286-9. [Google Scholar] |
| 42. | Otieno FO, Ndivo R, Oswago S, Ondiek J, Pals S, McLellan-Lemal E, et al. Evaluation of syndromic management of sexually transmitted infections within the Kisumu incidence cohort study. Int J STD AIDS 2014;25:851-9. [Google Scholar] |
| 43. | Lewis DA, Müller E, Steele L, Sternberg M, Radebe F, Lyall M, et al. Prevalence and associations of genital ulcer and urethral pathogens in men presenting with genital ulcer syndrome to primary health care clinics in South Africa. Sex Transm Dis 2012;39:880-5. [Google Scholar] |
| 44. | Suntoke TR, Hardick A, Tobian AA, Mpoza B, Laeyendecker O, Serwadda D, et al. Evaluation of multiplex real-time PCR for detection of Haemophilus ducreyi, Treponema pallidum, herpes simplex virus type 1 and 2 in the diagnosis of genital ulcer disease in the Rakai district, Uganda. Sex Transm Infect 2009;85:97-101. [Google Scholar] |
| 45. | Gomes Naveca F, Sabidó M, Amaral Pires de Almeida T, Araújo Veras E, Contreras Mejía Mdel C, Galban E, et al. Etiology of genital ulcer disease in a sexually transmitted infection reference center in Manaus, Brazilian Amazon. PLoS One 2013;8:e63953. [Google Scholar] |
| 46. | KularatneRS, Muller EE, Maseko DV, Kufa-Chakezha T, Lewis DA. Trends in the relative prevalence of genital ulcer disease pathogens and association with HIV infection in Johannesburg, South Africa, 2007-2015. PLoS One 2018;13:e0194125. [Google Scholar] |
| 47. | Wolday D, G-Mariam Z, Mohammed Z, Meles H, Messele T, Seme W, et al. Risk factors associated with failure of syndromic treatment of sexually transmitted diseases among women seeking primary care in Addis Ababa. Sex Transm Infect 2004;80:392-4. [Google Scholar] |
| 48. | Rahman M, Paz-Bailey G, Regoeng M, et al. Changing Patterns of STD in Botswana, 1993-2003. Abstract (No. 0334) presented at 15th Biennial Congress of the International Society for Sexually Transmitted Diseases Research (ISSTDR), Ottawa, Canada; 27-30 July, 2003. [Google Scholar] |
| 49. | World Health Organization. Guidelinesfor the Management of Sexually Transmitted Infections. Revised version 2003. Geneva:World Health Organization; 2003. [Google Scholar] |
| 50. | Burris H, Parkhurst J, Adu-Sarkodie Y, Mayaud P. Getting research into policy - Herpes simplex virus type-2 (HSV-2) treatment and HIV infection: International guidelines formulation and the case of Ghana. Health Res Policy Syst 2011;9 (Suppl 1):S5. [Google Scholar] |
| 51. | Sharma R, Prajapati S, Patel B, Kumar P. Evaluation of skill-oriented training on enhanced syndromic case management (ESCM) of reproductive tract infections / sexually transmitted infections (RTI/STIs) of care providers from three-tier healthcare system of Gujarat. Indian J Community Med 2016;41:183-9. [Google Scholar] |
| 52. | García PJ, Carcamo CP, Garnett GP, Campos PE, Holmes KK. Improved STD syndrome management by a network of clinicians and pharmacy workers in Peru: The PREVEN network. PLoS One 2012;7:e47750. [Google Scholar] |
| 53. | White RG, Moodley P, McGrath N, Hosegood V, Zaba B, Herbst K, et al. Low effectiveness of syndromic treatment services for curable sexually transmitted infections in rural South Africa. Sex Transm Infect 2008;84:528-34. [Google Scholar] |
Fulltext Views
15,330
PDF downloads
1,395




