Translate this page into:
Efficacy and safety of antivirals for Covid-19: A systematic review and meta-analysis
2 Department of Neurology, All India Institute of Medical Sciences, New Delhi, India
3 Department of Pulmonary Medicine, Critical Care and Sleep, All India Institute of Medical Sciences, New Delhi, India
4 Department of Medicine, All India Institute of Medical Sciences, New Delhi, India
5 Department of Haematology, All India Institute of Medical Sciences, New Delhi, India
6 Department of Medical Oncology, All India Institute of Medical Sciences, New Delhi, India
7 Department of Gastroenterology and Human Nutrition Unit, All India Institute of Medical Sciences, New Delhi, India
Corresponding Author:
Shalimar
Department of Gastroenterology and Human Nutrition Unit, All India Institute of Medical Sciences, New Delhi, India
India
drshalimar@yahoo.com
| How to cite this article: Prasad M, Elavarasi A, Madan K, Nischal N, Soneja M, Seth T, Sahoo RK, Sharma A, Garg P, S. Efficacy and safety of antivirals for Covid-19: A systematic review and meta-analysis. Natl Med J India 2020;33:222-231 |
Abstract
Background. Coronavirus disease 2019 (Covid-19) has led to a severe medical, social and economic crisis globally. Use of antivirals has given inconsistent results; thus systematic summaries of available evidence are required for any recommendations for treatment. We conducted a systematic review and meta-analysis on the use of antivirals for Covid-19.Methods. The databases we searched were—Medline, Embase, Cochrane CENTRAL and Medrxiv. Title/abstract screening, full-text screening and data abstraction were carried out in duplicate by two researchers. Pooled effect sizes and 95% confidence intervals (CI) were calculated using the Mantel-Haenszel method of random effects for meta-analysis.
Results. Twenty studies were found eligible for inclusion: 6 randomized controlled trials, 9 cohort studies and 5 case series. Moderate-quality evidence suggests a likely clinical benefit from the use of remdesivir in improving the number of recoveries (RR 1.18; 95% CI 1.07–1.31; I2 = 0%) and time to recovery in days (median –3.02; 95% CI –4.98 to –1.07; I2 = 97%). A possibility of lower mortality is suggested by low-quality evidence with remdesivir (RR 0.74; 95% CI 0.40–1.37, I2 = 58%). Moderate-quality evidence suggests no certain benefit of using lopinavir/ritonavir for Covid-19 compared to arbidol, lopinavir/ritonavir combined with arbidol or other medications used as controls.
Conclusion. Further evidence from randomized controlled trials is required for all antivirals to treat Covid-19. At present, remdesivir seems more promising than other antivirals.
Introduction
The pandemic of coronavirus disease 2019 (Covid-19), the illness caused by severe acute respiratory syndrome coronavirus- 2 (SARS-CoV-2), poses an unprecedented threat to human health. As of 28 May 2020, Covid-19 had resulted in more than 357 377 deaths worldwide.[1] The pandemic has left many parts of the world grappling with severe medical, social and economic crises.[2],[3]
The huge potential for adverse health consequences from this pandemic has led to a desperate need for interventions that can reduce morbidity and mortality. Research is ongoing to develop vaccines and identify therapeutics for Covid-19, including repurposing of medications.[4] However, research has so far been limited by short follow-up, exclusion of in-patients and small sample size.
The use of medications without proven efficacy may result in avoidable harm to patients, diversion of investment in other resources and erosion of public trust in the medical community.[5],[6] Antivirals used so far to treat Covid-19 have given inconsistent results. Thus, systematic summaries of available evidence are required to guide clinicians and policy-makers on use of antivirals. We conducted a systematic review and metaanalysis on the use of antivirals for Covid-19 to assess their efficacy and safety in patients.
Methods
We adhered to the statements of the preferred reporting items for systematic reviews and meta-analyses (PRISMA)[7] and meta-analyses and systematic reviews of observational studies in epidemiology (MOOSE)[8] and developed a protocol for systematic review.
Inclusion criteria
Type of participants: Studies on patients with severe and non-severe Covid-19.
Type of interventions: Studies that assessed any of the following drugs—ribavirin, remdesivir, umifenovir (arbidol), favipiravir and lopinavir–ritonavir
Type of outcomes: Studies that reported the following outcomes: (i) primary outcome: overall mortality and (ii) secondary outcomes: clinical recovery (as defined by authors), rate of admission to the intensive care unit (ICU), length of stay in ICU, length of hospital stay, need for mechanical ventilation, viral clearance and adverse events.
Type of studies: Randomized controlled trials (RCTs), cohort studies and case series. (Editorials, letters, news, reviews, expert opinions, case reports and studies without original data were excluded.)
Data sources and searches
We searched the following databases for articles published till 15 May 2020: Medline, Embase, Cochrane CENTRAL and PubMed (for articles not yet indexed in Medline). We also searched Medrxiv for pre-print articles.
Reference lists of retrieved articles and pertinent reviews were also searched for relevant articles. No language restriction was imposed. Appendix 1 [SUPPORTING:1] (available at www.nmji.in ) gives details of the search strategy.
Selection of studies
Titles and abstracts were screened in duplicate by two researchers (MP, S). Full texts of articles that either researcher considered potentially eligible were obtained. The eligibility of articles was determined from the full texts. Similarly, data were abstracted by two researchers independently and risk of bias was assessed. For all phases of the project, disagreement was resolved by discussion.
Data extraction
The data extracted from each study were author’s name, publication year, study design, setting/inclusion criteria, number of patients, patient characteristics, dose, duration and timing of antiviral therapy, co-medications, outcomes reported and method of adjustment (for cohort studies and case series).
Risk of bias assessment
Risk of bias was assessed using the modified version of the Cochrane Risk of Bias tool for RCTs, the revised version of Newcastle Ottawa Scale for Cohort studies and the Joanna Briggs Institute (JBI) checklist for case series.[9],[10],[11]
Data synthesis and statistical analysis
We calculated pooled risk ratio or risk difference and 95% CI using the Mantel–Haenszel method. The random effects model was used for meta-analysis. We carried out all statistical analyses using Review Manager 5.3. Heterogeneity was assessed using visual inspection of forest plot and the I2 statistic.
Grade
We used the GRADE methodology to rate certainty of evidence for each outcome as high, moderate, low or very low. We used detailed GRADE guidance to assess overall risk of bias, imprecision, inconsistency, indirectness and publication bias, and summarized results in an evidence profile.[12]
Results
Study selection
Our search yielded 1328 titles and abstracts—all were identified from the electronic database search. We excluded 1256 articles based on a review of the title and abstract, leaving 72 articles for full review. Of these, 52 were excluded—24 were case reports or reviews, 10 had an inappropriate study design, 15 reported irrelevant outcomes and 3 had an inappropriate comparison. Twenty studies were found eligible on full-text screening; 5 of these were pre-print articles that had not been peer-reviewed. These 20 studies were included in the systematic review[13],[14],[15],[16],[17],[18],[19],[20],[21],[22],[23],[24],[25],[26],[27],[28],[29],[30],[31],[32] and 12 studies were included in the meta-analysis [Figure - 1]. [13],[14],[15],[16],[17],[18],[19],[20],[27],[29],[30],[32]
 |
| Figure 1: PRISMA flow diagram for study selection[7] |
Study characteristics and estimates reported The studies included were 6 RCTs, 9 cohort studies and 5 case series.[13],[14],[15],[16],[17],[18],[19],[20],[21],[22],[23],[24],[25],[26],[27],[28],[29],[30],[31],[32] All studies included patients hospitalized with Covid- 19. Three trials[13],[14],[15] and two case series[25],[26] included mostly patients with severe Covid-19. The other three trials included patients with mild-to-moderate Covid-19.[16],[27],[32]
Two RCTs and two case series studied the drug remdesivir for Covid-19.[13],[14],[25],[26] Two retrospective cohort studies compared lopinavir/ritonavir to lopinavir/ritonavir along with arbidol,[17],[30] and three retrospective cohort studies and one RCT compared lopinavir/ritonavir to only arbidol.[18],[19],[20],[21],[22],[23],[24],[25],[26],[27],[28],[29],[30] Three RCTs and two retrospective cohort studies compared lopinavir/ritonavir to other medications used as controls.[15],[16],[19],[27],[30] One RCT each compared arbidol to placebo and favipiravir.[27],[32] One prospective cohort study compared lopinavir/ritonavir with favipiravir[21] [Table - 1] and [Table - 2].
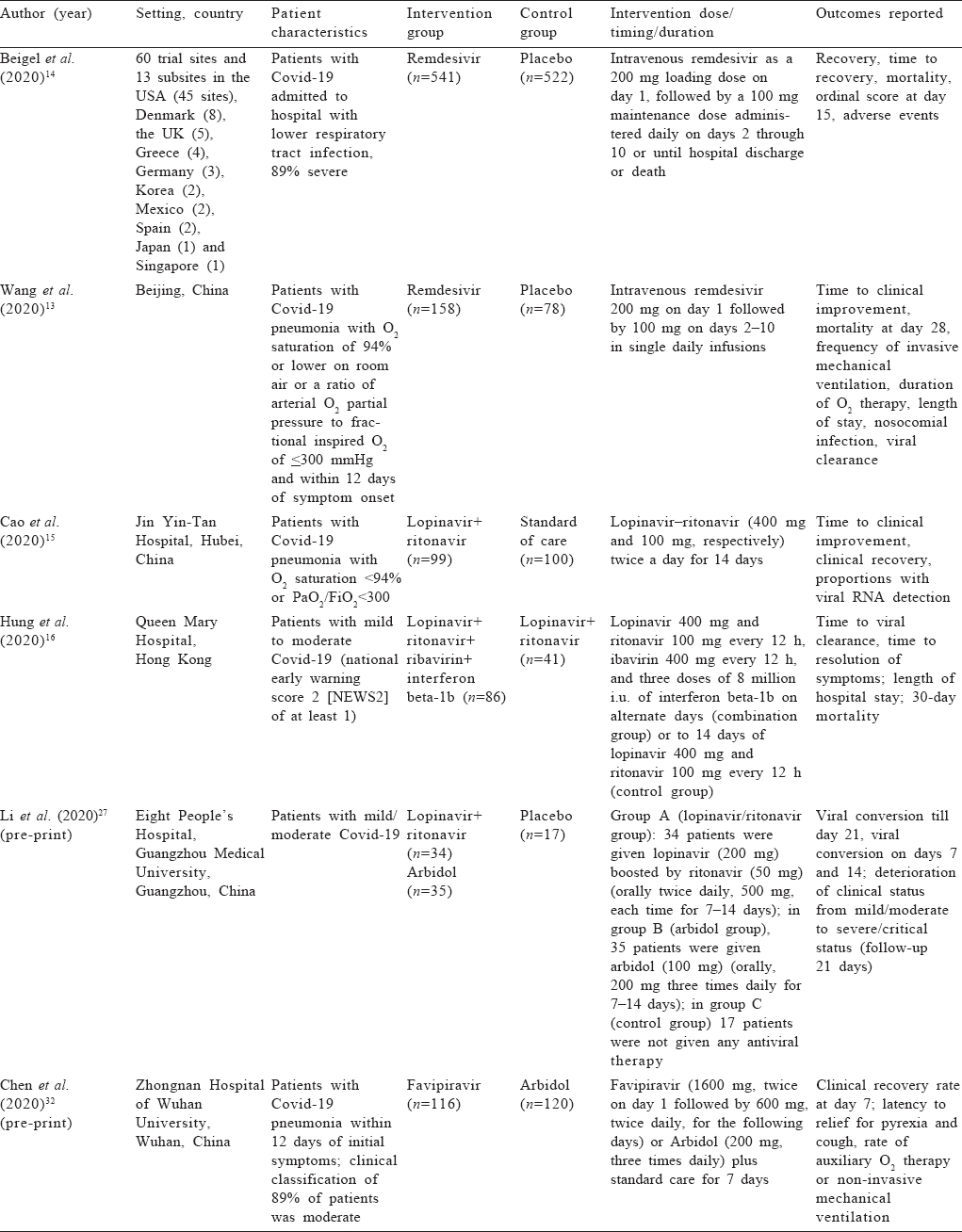
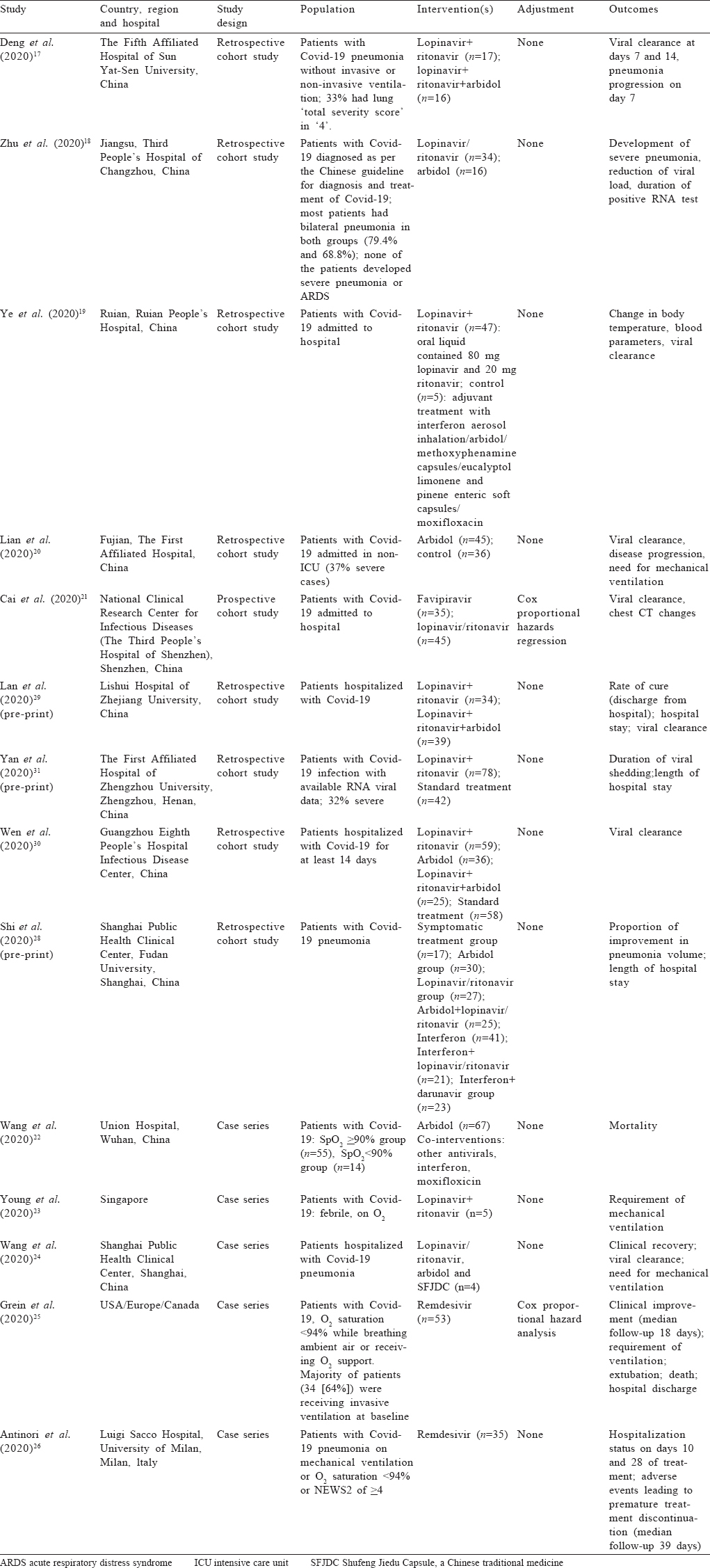
The outcomes reported by various studies are summarized in Appendix II [SUPPORTING:2] (available at www.nmji.in ).
Risk of bias assessment
All RCTs except one mentioned adequate generation of random sequence and blinding. However, only two trials mentioned adequate blinding. Missing outcome data were infrequent for all trials and two trials possibly had other sources of bias (Appendix III–I; [SUPPORTING:3] available at www.nmji.in ).
For all outcomes in the cohort studies, risk of bias was low for selection of exposed and non-exposed population and assessment of exposure. All cohort studies were assessed to have low risk of bias from outcome being present at the start of the study. Adequate adjustment and assessment of prognostic factors were carried out by only two studies. Follow-up was adequate for all outcomes in the cohort studies; however, only one study documented similar co-interventions in both groups (Appendix III–II; available at www.nmji.in ).
Most case series had well-defined criteria for inclusion and the condition measured in a standard, reliable way and clear reporting of the demographics of participants. However, most case series did not mention consecutive and complete inclusion of participants (Appendix III–III; available at www.nmji.in ).
Pooled effects of antivirals on safety and efficacy outcomes
Remdesivir versus placebo: Moderate-quality evidence from two RCTs[13],[14] (1295 participants) suggested a significantly greater number of clinical recoveries with remdesivir compared to placebo with no statistical heterogeneity (RR 1.18; 95% CI 1.07–1.31; I2=0%; [Figure - 2], [Table - 3]).
 |
| Figure 2: Effect of remdesivir on clinical recovery as compared to placebo |
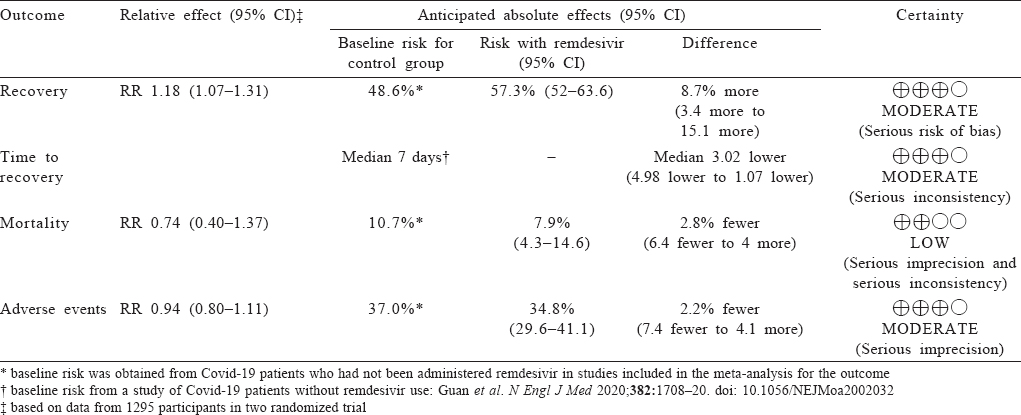
Moderate-quality evidence from two RCTs[13],[14] suggested a statistically significant shorter time to recovery with remdesivir when compared to placebo (mean deviation –3.02; 95% CI –4.98 to –1.07; I2=97%; [Figure - 3], [Table - 3]).
 |
| Figure 3: Effect of remdesivir on time to recovery as compared to placebo |
Low-quality evidence from two RCTs[13],[15] (1295 participants) raised the possibility of a reduction in mortality with remdesivir (RR 0.74; 95% CI 0.40–1.37, I2= 58%; [Figure - 4], [Table - 3]).
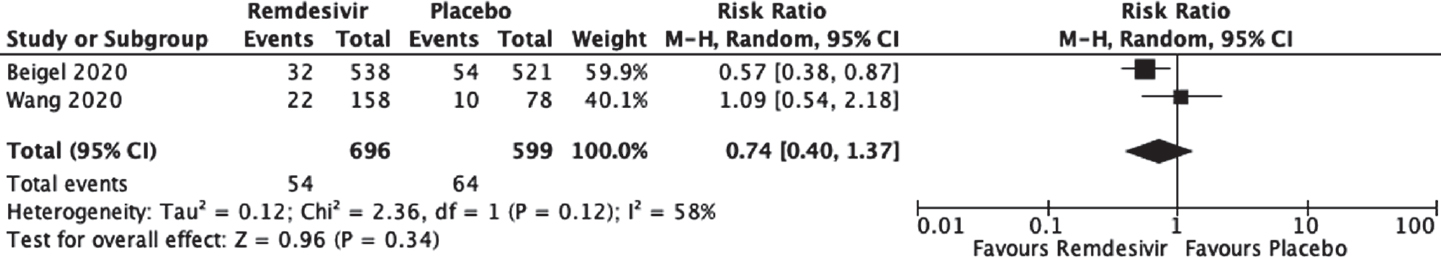 |
| Figure 4: Effect of remdesivir on mortality as compared to placebo |
Moderate-quality evidence from two RCTs[13],[14] (1295 participants) raised the possibility of no increase in adverse events due to remdesivir (RR 0.94; 95% CI 0.80–1.11; [Figure - 5], [Table - 3]).
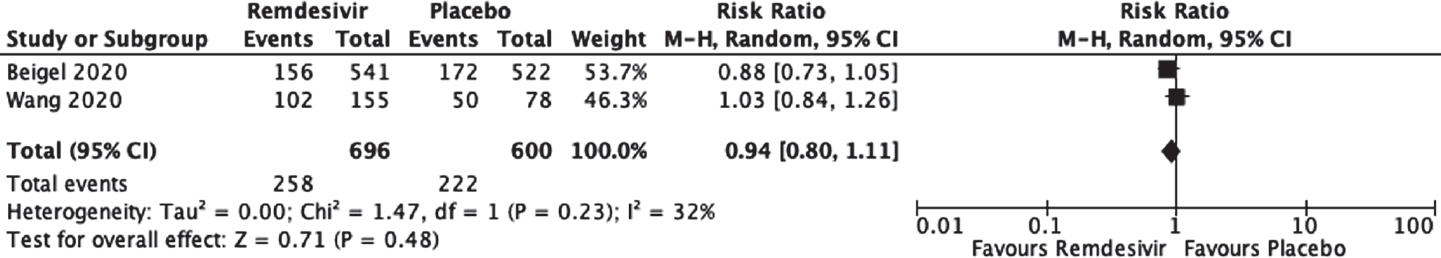 |
| Figure 5: Effect of remdesivir on adverse events as compared to placebo |
Lopinavir/ritonavir versus arbidol
One RCT[27] (69 participants) gave inconclusive results for viral clearance at day 7 (RR 0.95; 95% CI 0.51–1.78) and raised the possibility of an increase in adverse events (RR 2.47; 95% CI 0.97–6.26) with lopinavir/ritonavir compared to arbidol.
Very low-quality evidence from three cohort studies[18],[20],[30] (226 participants) suggested possible but uncertain effects of reduction in viral clearance at day 7 with lopinavir/ritonavir compared to arbidol (RR 0.65; 95% CI 0.35–1.22; [Figure - 6], [Table - 4]).
 |
| Figure 6: |

Lopinavir/ritonavir versus lopinavir/ritonavir combined with arbidol
One RCT[30] (84 participants) suggested a possible but uncertain reduction in viral clearance at day 7 with lopinavir/ritonavir compared to lopinavir/ritonavir plus arbidol (RR 0.51; 95% CI 0.17-1.51; [Figure - 7]).
 |
| Figure 7: |
Moderate-quality evidence from two cohort studies[17],[29] (106 participants) gave inconclusive results regarding a reduction in viral clearance at day 7 with lopinavir/ritonavir compared to lopinavir/ritonavir and arbidol (RR 0.73; 95% CI 0.22–2.45; [Figure - 7] and [Table - 5]).
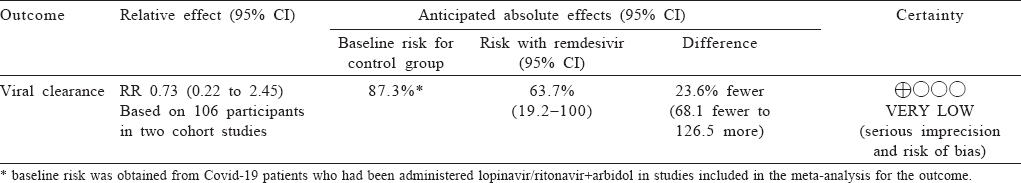
Lopinavir/ritonavir versus no antiviral
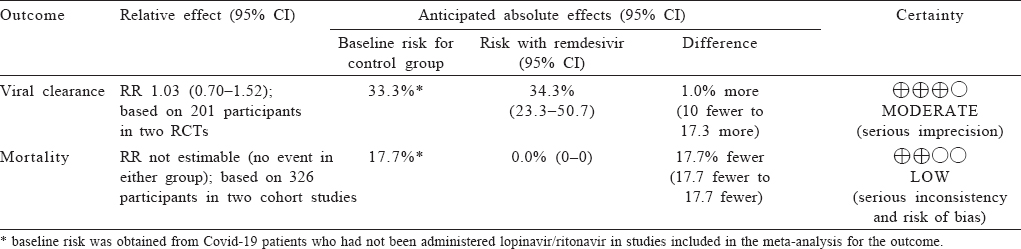
Moderate-quality evidence from two RCTs[15],[27] (201 participants) suggested no statistically significant reduction in viral clearance at day 7 with lopinavir/ritonavir compared to the control medication (RR 1.03; 95% CI 0.70–1.52; [Figure - 8] and [Table - 6]).
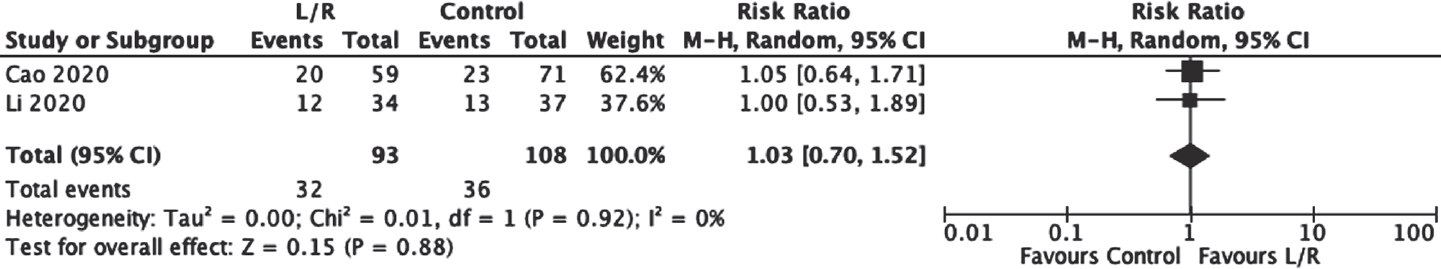 |
| Figure 8: Effect of lopinavir/ritonavir (L/R) on viral clearance as compared to control |
Favipiravir versus lopinavir/ritonavir
One cohort study[21] compared favipiravir to lopinavir/ritonavir and found a shorter viral clearance time for the favipiravir arm versus lopinavir/ritonavir: median 4 v. 11 days (p<0.001). The favipiravir arm also showed significant improvement in chest imaging compared with the control arm, with an improvement rate of 91.43% v. 62.22% (p=0.004).
Favipiravir versus arbidol
One RCT[32] that had not been peer-reviewed reported no statistically significant difference in the clinical recovery rate at day 7 in the favipiravir (61%) and arbidol (52%) groups.
Combination of interferon beta-lb, lopinavir–ritonavir, and ribavirin versus lopinavir/ritonavir alone
One RCT[16] compared the combination of interferon beta-1b, lopinavir–ritonavir, and ribavirin with lopinavir/ritonavir alone. The combination group had a significantly shorter median time from start of study treatment to negative nasopharyngeal swab (7 v. 12 days; hazard ratio 4.37; 95% CI 1.86-10.24, p=0.001).
Studies without a comparator
A case series[25] of patients who received remdesivir reported clinical improvement in 36 of 53 patients (68%) and mortality in 7 (13%). Another case series[26] of patients who received remdesivir reported clinical recovery at day 28 for 22 of 35 patients (63%).
In a series of 5 patients who received lopinavor/ritonavir, the fever resolved and supplemental oxygen requirement was reduced within 3 days for 3 patients, whereas the condition of 2 deteriorated with progressive respiratory failure. Four of 5 patients treated with lopinavir–ritonavir developed nausea, vomiting, and/or diarrhoea, and 3 developed abnormal liver function as shown by test results.[23]
Discussion
This systematic review provides moderate-quality evidence for the use of remdesivir in Covid-19 for improving the number of recoveries and time to recovery, and low-quality evidence for a possible reduction in mortality. Moderate-quality evidence suggests no benefit of using lopinavir/ritonavir in Covid-19 compared to arbidol, lopinavir/ritonavir combined with arbidol or other controls.
The clinical benefit in recovery with remdesivir, as reported by Beigel et al.,[14] is from the trial results published on the basis of a partial dataset. The data safety and monitoring board made a recommendation to unblind the results in view of the significant benefit. Thus, the imprecise estimates for the efficacy of remdesivir in reducing overall mortality, which this meta-analysis has yielded, are subject to change from the anticipated complete trial results.
In their preliminary report, Beigel et al. observed the reduction in time to recovery particularly for patients with severe disease (12 days in remdesivir recipients, compared with 18 days in recipients of placebo).[14] On the other hand, patients with mild or moderate disease experienced a time to recovery similar to those in the placebo group (5 days).
Wang et al.[13] have reported results that are consistent with those of Beigel et al. Their trial, which took place in China, could only enrol two-thirds of the intended patients since the outbreak had considerably diminished by that time, which explains the lack of precision in their estimates. The two trials reported mortality at different follow-up periods (14 days in Beigel et al. and 28 days in Wang et al.), which may have contributed to the heterogeneity.
Considering that both trials did observe mortality in a considerable proportion of participants receiving remdesivir (7%–8%), the question of discovering the best treatment for Covid-19 seems far from being resolved. It is also under investigation whether this mortality was due to the advanced stage of disease in these patients or they needed additional therapy. Additional concerns are the intravenous route of this drug, which precludes wider use. An endeavour to produce an oral formulation that can be taken by patients with moderate disease severity should be made.
Another trial compared the duration of remdesivir therapy and concluded no significant difference between a 5-day versus a 10-day course. The efficacy could not be determined due to the lack of placebo.[33]
The present systematic review has several strengths. It incorporates a robust and comprehensive search that covers three major databases in addition to unindexed and pre-print articles. The steps of screening and data abstraction, including risk of bias assessment, were conducted independently by two researchers. Lastly, we used the GRADE approach to rate the certainty in the evidence as very low, low, moderate or high. This enabled us to pay close attention to methodological issues such as imprecision, inconsistency and risk of bias.
The limitations of the present review are largely because only primary studies were included. The number of RCTs are so far, understandably, very few. Most cohort studies did not report using adjustment or assessment of prognostic factors, which resulted in rating down of the certainty in evidence.
There are, to the best of our knowledge, no previous systematic reviews that have incorporated all the studies included in the present review. A systematic review by Zhang et al[13] found that treatment with lopinavir–ritonavir showed no significant benefit in the rates of mortality and ARDS, which is consistent with our observations. Another systematic review[35] combined indirect evidence from SARS and MERS to some studies on Covid-19 and reported very low-quality evidence of a possible reduction in mortality from a combination of ribavirin and corticosteroids. A systematic review by Lui et al. did not study remdesivir and found only very low-quality evidence with little or no suggestion of benefit for most other treatments and outcomes in both non-severe and severe Covid-19.[36] Rios et al. included all the known antiviral treatments and antibodies for their potential treatment for Covid-19 and searched for all types of studies including preclinical (animal) studies. They summarized the results as inconclusive; however, they did not apply the GRADE approach.[37]
Limitations of the existing evidence make it imperative that researchers cooperate in conducting high-quality studies of efficacy, particularly RCTs.
In the light of these findings, remdesivir seems more promising than other medications. The medical community must commit to generating and implementing interventions with proven efficacy and safety, while discouraging the use of medications with very low-quality evidence.
| 1. | Covid-19 Map. Johns Hopkins coronavirus resource center. Available at https:/ /coronavirus.jhu.edu/map.html (accessed on 28 May 2020). [Google Scholar] |
| 2. | Fauci AS, Lane HC, Redfield RR. Covid-19—Navigating the uncharted. N Engl J Med 2020;382:1268-9. [Google Scholar] |
| 3. | Spinelli A, Pellino G. Covid-19 pandemic: Perspectives on an unfolding crisis. Br J Surg 2020;107:785-7. [Google Scholar] |
| 4. | Sanders JM, Monogue ML, Jodlowski TZ, Cutrell JB. Pharmacologic treatments for coronavirus disease 2019 (Covid-19): A review. JAMA 2020 Apr 13. doi: 10.1001/ jama.2020.6019. [Google Scholar] |
| 5. | Goodman JL, Borio L. Finding effective treatments for Covid-19: Scientific integrity and public confidence in a time of crisis. JAMA 2020 Apr 16. doi: 10.1001/ jama.2020.6434. [Google Scholar] |
| 6. | Juurlink DN. Safety considerations with chloroquine, hydroxychloroquine and azithromycin in the management of SARS-CoV-2 infection. CMAJ 2020 Apr 27;192:E450-3. doi: 10.1503/cmaj.200528. [Google Scholar] |
| 7. | Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M, et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev 2015;4:1. doi: 10.1186/2046-4053-4-1 [Google Scholar] |
| 8. | Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, et al. Meta-analysis of observational studies in epidemiology: A proposal for reporting. Meta-analysis of observational studies in epidemiology (MOOSE) group. JAMA 2000;283:2008-12. [Google Scholar] |
| 9. | Tool to Assess Risk of Bias in Cohort Studies. Available at https:// methods.cochrane.org/sites/methods.cochrane.org.bias/files/public/uploads/ Tool%20to%20Assess%20Risk%20of%20Bias%20in%20Cohort%20Studies.pdf (accessed on 29 May 2020). [Google Scholar] |
| 10. | Guyatt GH, Busse JW. Modification of Cochrane tool to assess risk of bias in randomized trials. Ottawa: Evidence Partners. Available at www.evidencepartners. com/resources/methodological-resources/ (accessed on 10 Jun 2020). [Google Scholar] |
| 11. | Moola S, Munn Z, Tufanaru C, Aromataris E, Sears K, Sfetcu R, et al. Systematic reviews of etiology and risk. In: Aromataris E, Munn Z (eds). Joanna Briggs Institute reviewer’s manual. The Joanna Briggs Institute; 2017. Available at https://reviewersmanual.joannabriggs.org/ (accessed on 10 Jun 2020). [Google Scholar] |
| 12. | Guyatt G, Oxman AD, Akl EA, Kunz R, Vist G, Brozek J, et al. GRADE guidelines: 1. Introduction—GRADE evidence profiles and summary of findings tables. J Clin Epidemiol 2011;64:383-94. [Google Scholar] |
| 13. | Wang Y, Zhang D, Du G, Du R, Zhao J, Jin Y, et al. Remdesivir in adults with severe Covid-19: A randomised, double-blind, placebo-controlled, multicentre trial. Lancet 2020;395:1569-78. [Google Scholar] |
| 14. | Beigel JH, Tomashek KM, Dodd LE, Mehta AK, Zingman BS, Kalil AC, et al. Remdesivir for the treatment of Covid-19—preliminary report. N Engl J Med 2020 May 22;NEJMoa2007764. doi: 10.1056/NEJMoa2007764. [Google Scholar] |
| 15. | Cao B, Wang Y, Wen D, Liu W, Wang J, Fan G, et al. A trial of lopinavir-ritonavir in adults hospitalized with severe Covid-19. N Engl J Med 2020;382:1787-99. [Google Scholar] |
| 16. | Hung IF-N, Lung K-C, Tso EY-K, Liu R, Chung TW-H, Chu M-Y, et al. Triple combination of interferon beta-1b, lopinavir-ritonavir, and ribavirin in the treatment of patients admitted to hospital with Covid-19: An open-label, randomised, phase 2 trial. Lancet 2020;395:1695-704. [Google Scholar] |
| 17. | Deng L, Li C, Zeng Q, Liu X, Li X, Zhang H, et al. Arbidol combined with LPV/ r versus LPV/r alone against corona virus disease 2019: A retrospective cohort study. J Infect 2020;81:e1-e5. doi: 10.1016/j.jinf.2020.03.002 [Google Scholar] |
| 18. | Zhu Z, Lu Z, Xu T, Chen C, Yang G, Zha T, et al. Arbidol monotherapy is superior to lopinavir/ritonavir in treating Covid-19. J Infect 2020;81:e21-e23. doi: 10.1016/j.jinf.2020.03.060 [Google Scholar] |
| 19. | Ye X-T, Luo Y-L, Xia S-C, Sun Q-F, Ding J-G, Zhou Y, et al. Clinical efficacy of lopinavir/ritonavir in the treatment of coronavirus disease 2019. Eur Rev Med Pharmacol Sci 2020;24:3390-6. [Google Scholar] |
| 20. | Lian N, Xie H, Lin S, Huang J, Zhao J, Lin Q. Umifenovir treatment is not associated with improved outcomes in patients with coronavirus disease 2019: A retrospective study. Clin Microbiol Infect 2020;26:917-21. doi: 10.1016/j.cmi.2020.04.026. Epub 2020 Apr 25. [Google Scholar] |
| 21. | Cai Q, Yang M, Liu D, Chen J, Shu D, Xia J, et al. Experimental treatment with favipiravir for Covid-19: An open-label control study. Engineering (Beijing). 2020 Mar 18. doi: 10.1016/j.eng.2020.03.007. Epub ahead of print. [Google Scholar] |
| 22. | Wang Z, Yang B, Li Q, Wen L, Zhang R. Clinical features of 69 cases with coronavirus disease 2019 in Wuhan, China. Clin Infect Dis 2020 Mar 16;ciaa272. doi: 10.1093/cid/ciaa272. [Google Scholar] |
| 23. | Young BE, Ong SWX, Kalimuddin S, Low JG, Tan SY, Loh J, et al. Epidemiologic features and clinical course of patients infected with SARS-CoV-2 in Singapore. JAMA 2020;323:1488-94. [Google Scholar] |
| 24. | Wang Z, Chen X, Lu Y, Chen F, Zhang W. Clinical characteristics and therapeutic procedure for four cases with 2019 novel coronavirus pneumonia receiving combined Chinese and western medicine treatment. Biosci Trends 2020;14:64-8. [Google Scholar] |
| 25. | Grein J, Myers RP, Brainard D. Compassionate use of remdesivir in Covid-19. Reply. N Engl J Med 2020;382:e101. [Google Scholar] |
| 26. | Antinori S, Cossu MV, Ridolfo AL, Rech R, Bonazzetti C, Pagani G, et al. Compassionate remdesivir treatment of severe Covid-19 pneumonia in intensive care unit (ICU) and non-ICU patients: Clinical outcome and differences in post-treatment hospitalisation status. Pharmacol Res 2020;158:104899. doi:10.1016/ j.phrs.2020.104899 [Google Scholar] |
| 27. | Li Y, Xie Z, Lin W, Cai W, Wen C, Guan Y, et al. An exploratory randomized controlled study on the efficacy and safety of lopinavir/ritonavir or arbidol treating adult patients hospitalized with mild/moderate Covid-19 (ELACOI). medRxiv. Available at www.medrxiv.org/content/10.1101/2020.03.19.20038984 v2 (accessed on 29 May 2020). [Google Scholar] |
| 28. | Shi X, Lu Y, Li R, Tang Y, Shi N, Song F, et al. Evaluation of antiviral therapies for coronavirus disease 2019 (Covid19) pneumonia in Shanghai, China. J Med Virol Available at https://onlinelibrary. wiley. com/doi/abs/10.1002/jmv.25893 (accessed on 29 May 2020). [Google Scholar] |
| 29. | Lan X, Shao C, Zeng X, Wu Z, Xu Y. Lopinavir-ritonavir alone or combined with arbidol in the treatment of 73 hospitalized patients with Covid-19: A pilot retrospective study. medRxiv. Available at www.medrxiv.org/content/10.1101/ 2020.04.25.20079079v1 (accessed on 29 May 2020). [Google Scholar] |
| 30. | Wen CY, Xie ZW, Li YP, Deng XL, Chen XT, Cao Y, et al. Real-world efficacy and safety of lopinavir/ritonavir and arbidol in treating with Covid-19: An observational cohort study [Chinese]. ZhonghuaNeiKe Za Zhi 2020;59:E012. [Google Scholar] |
| 31. | Yan D, Liu XL, Zhu Y, Huang L, Dan B, Zhang G, et al. Factors associated with prolonged viral shedding and impact of Lopinavir/Ritonavir treatment in patients with SARS-CoV-2 infection. medRxiv. Available at www.medrxiv.org/content/ 10.1101/2020.03.22.20040832v2 (accessed on 29 May 2020). [Google Scholar] |
| 32. | Chen C, Zhang Y, Huang J, Yin P, Cheng Z, Wu J, et al. Favipiravir versus arbidol for Covid-19: A randomized clinical trial. medRxiv. Available at www.medrxiv.org/ content/10.1101/2020.03.17.20037432v4 (accessed on 29 May 2020). [Google Scholar] |
| 33. | Goldman JD, Lye DCB, Hui DS, Marks KM, Bruno R, Montejano R, et al. Remdesivir for 5 or 10 days in patients with severe Covid-19. N Engl J Med 2020 Available at www.nejm.org/doi/full/10.1056/NEJMoa2015301 (accessed on 5 Jun 2020). [Google Scholar] |
| 34. | Zhang L, Liu Y. Potential interventions for novel coronavirus in China: A systematic review. J Med Virol 2020;92:479-90. [Google Scholar] |
| 35. | Zhong H, Wang Y, Zhang Z-L, Liu Y-X, Le K-J, Cui M, et al. Efficacy and safety of current therapeutic options for Covid-19—lessons to be learnt from SARS and MERS epidemic: A systematic review and meta-analysis. Pharmacol Res 2020;157:104872. doi: 10.1016/j.phrs.2020.104872 [Google Scholar] |
| 36. | Liu W, Zhou P, Chen K, Ye Z, Liu F, Li X, et al. Efficacy and safety of antiviral treatment for Covid-19 from evidence in studies of SARSCoV-2 and other acute viral infections: A systematic review and meta-analysis. CMAJ 2020;192:E734-E744. doi: 10.1503/cmaj.200647 [Google Scholar] |
| 37. | Rios P, Radhakrishnan A, Antony J, Thomas SM, Muller M, Straus SE, Tricco AC. Effectiveness and safety of antiviral or antibody treatments for coronavirus: A rapid review. medRxiv. Available at www.medrxiv.org/content/10.1101/ 2020.03.19.20039008v2 (accessed on 10 Jun 2020). [Google Scholar] |
Fulltext Views
1,712
PDF downloads
311




