Translate this page into:
Performance of equations for estimation of 24-hour urinary sodium from morning fasting urine samples in a multisite study in India
Correspondence to LAKSHMY RAMAKRISHNAN; lakshmy_ram@yahoo.com
To cite: Krishnan A, Amarchand R, Tarik M, Salve HR, Raghav P, Patro BK, et al. Performance of equations for estimation of 24-hour urinary sodium from morning fasting urine samples in a multisite study in India. Natl Med J India 2022;35:4–10.
Abstract
Background
Dietary salt intake is an important modifiable risk factor for cardiovascular diseases. Estimation of 24-hour salt intake using morning urine samples needs to be validated in the Indian context. We examined the performance of INTERSALT, Tanaka and Kawasaki equations for the estimation of 24-hour urinary sodium from morning fasting urine (MFU) samples.
Methods
We enrolled 486 adults aged 18–69 years from four regions of India with equal rural/urban and sex representation to provide 24-hour urine samples. The next day, a MFU sample was obtained. Based on the volume and sodium content of the 24-hour urine sample, 24-hour sodium excretion (reference method) was calculated. Sodium levels in the MFU samples were measured along with other parameters required, and the above equations were used to estimate 24-hour urinary sodium levels. Intraclass correlation coefficient (ICC) was used to assess the degree of agreement between the estimates from the reference method and the three equations. Bland–Altman (BA) plots were used to identify systematic bias and limits of agreement. A difference of 1 g of salt (0.39 g of sodium) between the mean salt intake by 24-hour urine and as estimated by equations was considered acceptable.
Results
A total of 346 participants provided both the samples. The mean (SD) daily salt intake estimated by the 24-hour urine sample method was 9.9 (5.8) g. ICC was low for all the three equations: highest for Kawasaki (0.16; 95% CI 0.05–0.26) and least for Tanaka (0.12; 0.02–0.22). Only Tanaka equation provided estimates within 1 g of measured 24-hour salt intake (–0.36 g). BA plots showed that as the mean values increased, all the three equations provided lower estimates of salt intake.
Conclusion
Tanaka equation provided acceptable values of 24-hour salt intake at the population level. However, poor performance of all the equations highlights the need to understand the reasons and develop better methods for the measurement of sodium intake at the population level.
INTRODUCTION
Dietary salt intake is strongly associated with blood pressure and is an important risk factor for cardiovascular diseases. The Global Burden of Disease study estimated that high salt intake was the tenth highest risk factor of disease burden, and diets high in sodium were responsible for 127.5 million disability-adjusted life years.1 Reducing population salt intake has been identified as one of the most cost-effective interventions for reducing the non-communicable disease (NCD) burden.2 The mean 24-hour salt intake is one of the indicators identified as a part of the global NCDs monitoring framework.3 A major challenge is the difficulty to measure and monitor change at the population level.
Dietary surveys are not considered a reliable method of measuring salt intake due to the problem of underestimation. The ‘gold-standard’ approach to assessing mean population salt intake is to obtain urine samples over 24 hours on a representative sample of the population and use them to estimate daily salt intake.4 However, this approach poses challenges of operational feasibility, skills and resources required for national-level health surveys in low- and middle-income countries.5 Therefore, if dietary salt intake is to be monitored as a regular indicator under NCD surveillance, feasible alternatives need to be explored.
Sodium estimation in a ‘spot’ urine sample is a widely studied option. It has been shown to have a good correlation with 24-hour sodium excretion at the population level in many studies. INTERSALT, Tanaka and Kawasaki are the three most commonly used mathematical equations for the estimation of 24-hour sodium excretion from spot sample sodium values.6–8 The World Health Organization (WHO) in 2013 recommended that spot urine specimens may be used to estimate mean population salt intake (using the INTERSALT equation [without potassium]) while continuing the search for better ways of estimating 24-hour salt intake from spot urine specimens.9,10 There is no clear guidance about the timing of spot urine as studies have used different spot samples—casual, early morning, mid-day and morning fasting samples.11,12 Significant differences in urinary sodium excretion by the timing of collection have been reported by studies.13 Studies comparing the validity of the three equations have identified limitations with all the three equations, and no firm recommendation has been made regarding the choice of equation for a given population.14–16 A study of 11 countries recommended that in a diverse population, Kawasaki formula was the most valid (intraclass correlation coefficient [ICC] of 0.71) and the least biased method of estimating 24-hour sodium excretion from a single morning fasting urine (MFU) sample and is suitable for population studies.17 A review recommended that in those providing spot urine, 24-hour urine collections should also be done in a sub-sample to enable the development of valid equations that can provide a robust mean population estimate of sodium intake from spot collections.18
India identified mean population daily salt intake as one of its indicators with targets in its national NCD monitoring framework in 2013.19 Thereafter, at the behest of the Ministry of Health and Family Welfare, Government of India, a National NCD Monitoring Survey (NNMS) was planned by the Indian Council of Medical Research, in which urinary sodium estimation was included in a sub-sample of adults aged 18–69 years. The Technical Working Group (TWG) of the NNMS reviewed an earlier study covering two parts of India, which used the morning spot (non-fasting) sample, and found that Kawasaki equation (ICC of 0.4) performed the worst among the equations tested and overestimated daily intake by 3–4 g.20 It was, therefore, recommended by the TWG to carry out a multisite sub-study to evaluate the performance of the three equations for the estimation of 24-hour urinary sodium from spot urine samples for India. As the NNMS planned to link spot urine collection with fasting blood sugar estimation, it was decided to use the morning fasting sample for urinary sodium estimation. The results of this study would inform the larger NNMS survey on the calculation of urinary sodium and salt intake.
METHODS
The study was conducted at four sites in four regions namely Jodhpur (West), Delhi (North), Chennai (South) and Bhubaneswar (East), in India in early 2017. At each site, one village and one urban colony were selected based on geographical access. Institutional ethics committees of the All India Institute of Medical Sciences, New Delhi (the coordinating site), as well as of the other three institutions approved the study. Written informed consent was obtained from each study participant before enrolment. The WHO India country office funded the study.
Adults aged 18–69 years, willing to participate in the study by providing 24-hour urine sample collection, were identified. Pregnant women, patients with a known history of heart or kidney failure or liver disease, those who are taking therapy with diuretics within the preceding 2 weeks and those with any gastrointestinal surgery were excluded from the study. In the study cluster, 60 individuals were selected based on availability and consent to provide 24-hour urinary sample. Quota sampling was used to ensure uniform distribution of participants in each of the four strata: age (18–44; 45–69 years) and sex (men/ women).
A semi-structured pretested interview schedule was used to collect baseline sociodemographic information, data on tobacco and alcohol use, knowledge and practices regarding dietary salt intake as well as personal medical history including drug treatment for hypertension. Baseline physical examination including blood pressure and anthropometric (weight and height) measurements were made using standard equipment and protocol. The protocol used to collect 24-hour urine and morning fasting sample collection was explained by trained investigators in local language. The respondents were provided detailed written instructions in addition to verbal instructions. For illiterate respondents, one of the literate family members was given the written instructions in addition to verbal instructions to the respondent. At each site, two medical social workers (one man and one woman) and two field attendants were trained on the procedures of spot and 24-hour urine sample collection.
The participants were asked to record the time of the start and end of 24-hour urine collection and the time of spot urine collection and report any missed collections during the 24-hour period. During the collection period, the participants were instructed to maintain a normal diet and their usual life routine. They were asked to collect a 24-hour urine sample as described in PAHO protocols on a day most convenient to them.4 The participants were asked to choose a day when they will be mostly at home or only going out for a short time. In the morning of the start of the 24-hour period, the participants were told to void the bladder and note the time. This ‘first-pass urine’ was to be discarded. All urine passed thereafter was collected in the container provided, including the first urine of the following morning, with the final time recorded. A MFU sample (30 ml) was also collected from the participant on the next day in a separate sterile sample bottle provided to them. Duration of fasting was also recorded at the time of sample collection. If the identified person could not complete the protocol, someone replaced him or her from the same house or a neighbouring one.
24-hour urine samples were collected in 5 L containers (with no additives) by field researchers on the day of completion. After shaking for homogenization, the volume of collection was measured and 30 ml was transferred to a sterile plastic bottle, while the rest was discarded. These were transported to the laboratory of the local institutes on the same day at 4 °C using cold boxes. At the local laboratory, two 1.5 ml aliquots were drawn and stored at –20 °C, which were then transferred to a central laboratory at AIIMS, New Delhi, in dry ice for analysis. Sodium and potassium content in the urine was determined through ion selective electrode (indirect) method by an auto-analyser (AU680, Beckman Coulter Diagnostics), and creatinine content was determined by Jaffe’s kinetic method using kits specific for urine on the auto-analyser. The validity of the urine sample was determined by the creatinine values. Standard internal quality assurance protocols were followed. The laboratory at AIIMS, New Delhi is a part of an international quality control system (Randox External Quality Assessment Scheme).
Statistical analysis
A sample size of 288 was required based on the assumption of ICC of 0.4 and an acceptable width of 0.2 for 95% CI.21 Hence, 120 participants at each site were to be included to adjust for dropouts, totalling 480 study participants. Only participants for whom both a valid 24-hour urine sample (>20 or <28 hours or >500 ml collection of urine) and a MFU sample were available were included in the analysis. Data were entered in Microsoft Excel, and analysis was carried out using STATA version 14. The 24-hour sodium excretion value (mmol/day) was calculated as the concentration of sodium in the urine (mmol/L) multiplied by the urinary volume (L/day) multiplied by a time adjustment factor to standardize to a 24-hour period. The conversion of sodium (mmol/day) to sodium (mg/day) was made by multiplying by 23. The conversion of sodium (g/day) to salt (g/day) was made by multiplying the sodium value by 2.52. All the three, Kawasaki, Tanaka and INTERSALT (with and without potassium), equations were used to estimate 24-hour salt intake using spot sample results for sodium, potassium and creatinine (Box 1). Sodium intake was expressed as mg/day, and the mean and SD were calculated. Daily salt intake as estimated from these equations was compared with salt intake measured from the 24-hour sample values. A difference of 1 g of salt (0.39 g of sodium) between the 24-hour urine and as estimated by equations was considered acceptable. Cases with extreme values for sodium, potassium and creatinine were considered as outliers and removed from the analysis.
| Equation | |
|---|---|
| Kawasaki equation | 23×[16.3× √ {Spot Na (mmol/l)/(Spot Cr (mg/dl)×10)×predicted 24-h urinary Cr (mg/day)}], where predicted Cr (mg/day) for women is as follows: [–4.72×age (years)+8.58× weight (kg)+5.09×height (cm)–74.5], for men it is [–12.63×age (years)+15.12×weight (kg)+7.39× height (cm)–79.9] |
| Tanaka equation | 23×[21.98×XNa0.392], where XNa=[spot Na (mmol/l)/ [spot creatinine (mg/dl)× 10]]×[Predicted 24-h urinary Cr (mg/day)]; and Predicted Cr (mg/day)= [–2.04×age (years)]+[14.89×weight (kg)]+[16.14×height (cm)] –2244.45. |
| Intersalt equation | For men: 23×{25.46+[0.46×spot Na (mmol/l)]– [2.75×spot Cr (mmol/l)]– [0.13×spot K (mmol/ l)]+[4.10×BMI (kg/m2)]+[0.26×age (years)]}, and For women: 23×{5.07+[0.34×spot Na (mmol/l)]– [2.16×spot Cr (mmol/l)]–[0.09×spot K (mmol/l)] + [2.39×BMI (kg/m2)] + [2.35×age (years)]– [0.03×age2(years)]}. |
| INTERSALT without potassium | Male:23×(23.51+[0.45×spot Na (mmol/L)]–[3.09× spot Cr (mmol/L)]+[4.16×BMI (kg/m2)]+[0.22×age (years)]) Female : 23×(3.74+[0.33×spot Na (mmol/L)]–[2.44×spot Cr (mmol/L)]+[2.42×BMI (kg/m2)]+[2.34×age (years)–[0.03×age2 (years)]) |
Pearson’s correlation coefficient was estimated between sodium levels in MFU samples and with 24-hour urinary sodium and between estimated sodium from spot urine MFU samples (using the equations of Kawasaki, Tanaka and INTERSALT) and the measured 24-hour urine. The correlations between estimated sodium excretion from MFU samples and measured 24-hour urine were also quantified by ICC using a two-way mixed-effects model for absolute agreement. Bland–Altman (BA) plot analysis was done by plotting the mean of the measured and estimated 24-hour sodium excretion values, and the difference between the two values of estimated sodium excretion for the same individual (by all the three equations) and mean bias (predicted–measured) and statistical limits of agreement were estimated. Further details of these standard procedures are provided in the literature.20,22
RESULTS
A total of 486 participants were included in the study with almost similar numbers in all the four sites. Of the 486 individuals, 140 (28.8%) were excluded from the analysis due to the reasons mentioned (Fig. 1) or damage to the samples during transport to the laboratory in New Delhi. The sociodemographic characteristics of these 346 individuals are given in Table I. The study participants were almost equally divided into rural and urban, man and woman, and age brackets 18–44 versus 45–69 years. The majority of respondents (85%) had completed more than secondary schooling. The overall prevalence of overweight (body mass index [BMI] >25 kg/m2) was 39.8% with significant differences between the four sites. The Chennai site had the highest prevalence of overweight (53.3%) and Bhubaneswar had the least (28.5%). Almost half of the sample population was adding extra salt to the plate at the time of eating, with the least being at Chennai (37.1%). The highest mean blood pressure values were reported from Delhi (systolic 134.0 mmHg and diastolic of 85.1 mmHg) and lowest from Bhubaneswar (systolic 117.9 mmHg and diastolic 79.2 mmHg).
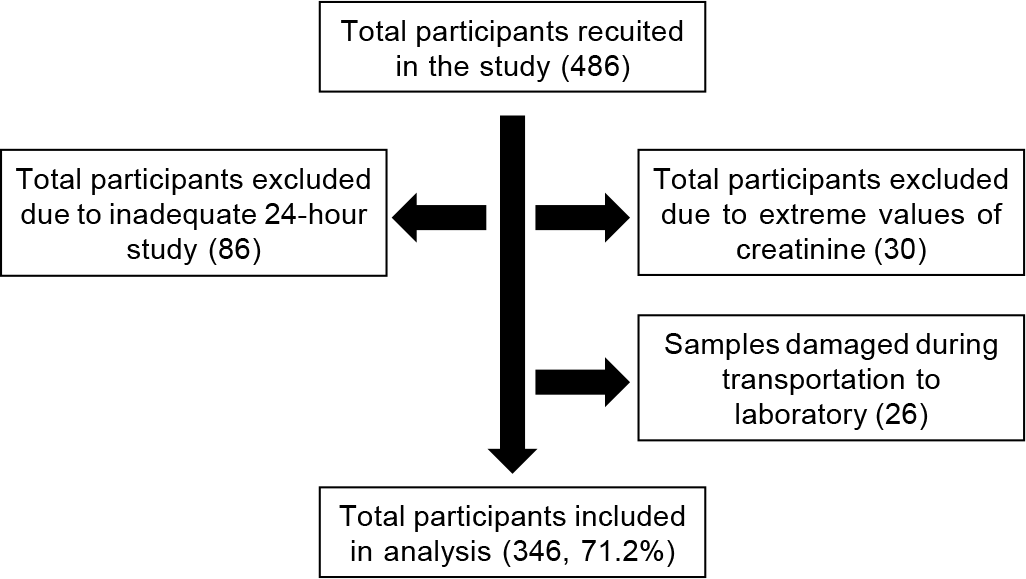
- Flow of the study participants
| Characteristic | Jodhpur (n=65) | Delhi (n=100) | Chennai (n=90) | Bhubaneswar (n=91) | Combined (n=346) |
|---|---|---|---|---|---|
| Setting, n (%) | |||||
| Rural | 27 (41.5) | 50 (50.0) | 51 (56.6) | 46 (50.5) | 174 (50.2) |
| Urban | 38 (58.4) | 50 (50.0) | 39 (43.3) | 45 (49.4) | 172 (49.7) |
| Sex, n (%) | |||||
| Man | 46.1 | 52.0 | 50.0 | 46.1 | 48.8 |
| Woman | 53.8 | 48.0 | 50.0 | 53.8 | 51.1 |
| Age between 18 and 44 years (%) | 49.2 | 4 6 | 5 0 | 50.5 | 48.8 |
| Percentage who completed more than secondary schooling | 80.0 | 84.2 | 90.4 | 83.5 | 85.0 |
| Percentage who were overweight or obese (BMI >25) | 44.6 | 35.0 | 53.3 | 28.5 | 39.8 |
| Percentage who added extra salt to food during orbefore eating | 61.5 | 54.0 | 37.1 | 54.9 | 49.7 |
| Mean (SD) blood pressure (mmHg) | |||||
| Systolic | 125.4 (1.9) | 134.0 (2.1) | 126.3 (2.0) | 117.9 (1.8) | 126.2 (1.0) |
| Diastolic | 82.6 (1.2) | 85.1 (1.0) | 84.0 (1.2) | 79.2 (3.9) | 82.8 (1.2) |
| Mean (range) 24-h urine collected (L) | 2.3 (0.5–4) | 1.3 (0.5–5) | 1.7 (0.5–4.4) | 2.4 (0.5–5) | 1.9 (0.5–5) |
| Mean (SD) daily sodium intake by 24-hour urine method (mg) | 4269.4 (368.6) | 3191.9 (170.1) | 4014.3 (225.6) | 4373.2 (257.4) | 3918.9 (125.5) |
| Mean (SD) creatinine levels in 24-hour urine sample (mg/dl) | 74.3 (7.3) | 62.3 (3.2) | 67.3(4.3) | 48.9 (5.3) | 62.4 (2.5) |
| Mean (SE) potassium level in 24-hour urine sample (mmol/dl) | 32.6 (2.9) | 32.3 (1.6) | 23.6 (1.4) | 19.4 (1.8) | 26.7 (0.9) |
Mean salt intake can be estimated by multiplying sodium levels by 2.52 SD standard deviation BMI body mass index
On average, the participants collected 1.9 L of urine in 24 hours, with the least being in Delhi (1.3 L) and highest in Bhubaneswar (2.4 L). The mean daily sodium intake estimated by the 24-hour urine sample method (reference method) was 3.9 g (equivalent to salt intake of 9.9 g). Delhi had the lowest sodium intake (3.2 g; equivalent to 8 g of salt) and Bhubaneswar reported the highest sodium intake (4.4 g; equivalent to 11 g of salt). There were major differences in the creatinine and potassium levels between the four sites as well.
The performance of the three equations using MFU sample sodium levels was compared with the reference method (Table II). INTERSALT equation underestimated 24-hour sodium excretion by 569 mg, whereas the other two equations overestimated sodium excretion, with Tanaka providing the closest value to the measured 24-hour sodium excretion (144.4 mg). The BA plots also revealed a systematic bias as all the three equations provided lower estimates of salt intake as the mean values increased (Fig. 2). Pearson’s correlation coefficient was highest for INTERSALT (0.23) and least for Kawasaki (0.17). ICC was highest for Kawasaki (0.16; 95% CI 0.05–0.26) and least for Tanaka (0.12; 0.02–0.22). When converted to salt intake (*2.52) and classified as proportion of population with intake more than or equal to the recommended intake (<5 g/day), the 24-hour urine method estimated it as 81.2%. Others estimated it much higher, including INTERSALT which had a lower mean sodium intake. There was not much difference in the estimate generated by INTERSALT equation with or without potassium.
| Item | Measured 24-hour excretion (mg) | Kawasaki equation (mg) | INTERSALT equation (mg) | Tanaka equation (mg) | INTERSALT equation without potassium (mg) | |
|---|---|---|---|---|---|---|
| Mean (SD) sodium level (mg/day) | 3918.9 (2336.2) | 5692.3 (1607.2) | 3349.3 (701.8) | 4063.3 (936.9) | 3312.9 (694.3) | |
| Range of sodium levels (mg/day) | 563–13 524 | 1948–15 140 | 1555–5780 | 1618–8934 | 1523–5720 | |
| Pearson’s correlation coefficient | Reference | 0.17 (0.064–0.279) | 0.23 (0.133–0.344) | 0.18 (0.071–0.288) | 0.23 (0.121–0.346) | |
| ICC (95% CI) | Reference | 0.159 (0.054–0.260) | 0.129 (0.024–0.231) | 0.122 (0.017–0.224) | 0.128 (0.023–0.230) | |
| Mean bias (95% CI) | Reference | 1800 (1500–2000) | –569.5 (–810.3 – –328.8) | 144.3 (105.1–393.7) | –605.9 (–846.6 – –365.3) | |
| Proportion estimated to have a daily intake of salt >5 g/day | 81.2 | 99.7 | 99.4 | 99.4 | 99.4 | |
ICC intraclass correlation coefficient CI confidence interval SD standard deviation

- Bland–Altman (BA) plots for comparison of sodium level estimation by 24-hour urine collection with that estimated from different equations
| Characteristic | Measured 24-hour excretion (mg) | Kawasaki equation (mg) | INTERSALT equation (mg) | Tanaka equation (mg) | INTERSALT equation without potassium (mg) |
|---|---|---|---|---|---|
| Site (95% CI) | |||||
| Rural | 4208.3 | 0.134 | 0.071 | 0.101 | 0.073 |
| (3856.2–4560.3) | (–0.014–0.277) | (–0.078–0.217) | (–0.048–0.246) | (–0.076–0.219) | |
| Urban | 3623.3 | 0.162 | 0.183 | 0.122 | 0.179 |
| (3284.3–3968.2) | (0.013–0.304) | (0.035–0.323) | (–0.028–0.266) | (0.030–0.319) | |
| Sex (95% CI) | |||||
| Man | 4115.1 | 0.181 | 0.136 | 0.138 | 0.138 |
| (3752.4–4477.6) | (0.031–0.323) | (–0.014–0.281) | (–0.012–0.283) | (–0.013–0.282) | |
| Woman | 3731.7 | 0.124 | 0.081 | 0.116 | 0.079 |
| (3396.5–4066.9) | (–0.023–0.267) | (–0.067–0.226) | (–0.032–0.258) | (–0.069–0.224) | |
| Site | |||||
| Jodhpur | 4269.4 | 0.050 | 0.175 | 0.037 | 0.169 |
| (3544.3–4994.3) | (–0.195–0.289) | (–0.070–0.400) | (–0.207–0.277) | (–0.076–0.396) | |
| Delhi | 3191.9 | 0.232 | 0.206 | 0.194 | 0.208 |
| (2857.2–3526.6) | (0.038–0.409) | (0.011–0.386) | (–0.001–0.376) | (0.013–0.388) | |
| Chennai | 4014.3 | 0.224 | 0.172 | 0.180 | 0.175 |
| (3570.5–4458.2) | (0.019–0.411) | (–0.035–0.365) | (–0.027–0.372) | (–0.033–0.367) | |
| Bhubaneswar | 4373.2 | 0.067 | 0.064 | 0.053 | 0.064 |
| (3866.8–4879.5) | (–0.140–0.269) | (–0.143–0.265) | (–0.154–0.255) | (–0.143–0.265) |
CI confidence interval
A sub-analysis of the estimated salt intake indicated rural– urban differences (4.2 v. 3.6 g sodium/day) and gender differences (4.1 v. 3.7 g sodium/day), both of which were not significant. All the three equations performed better among men and among individuals from urban areas.
DISCUSSION
We assessed the performance of the three most commonly used equations to predict 24-hour sodium excretion (proxy for intake) at four sites in India. All the three equations had poor ICC values. Kawasaki had the highest mean bias. INTERSALT underestimated 24-hour urinary excretion and Tanaka overestimated it, but only Tanaka prediction had its 95% CI of the mean differences within the proposed 1 g limit.
Comparison of studies done to answer the question of performance of different equations for predicting 24-hour urinary excretion from spot samples are beset with multiple methodological problems—choice of spot sample (fasting, morning and afternoon), timing (within 24-hour or outside the 24-hour window) and use of statistical methods (correlation coefficient, ICC, BA plots and validity parameters).14–18 There are no standard values for acceptable reliability using ICC.20 A low ICC reflects the low degree of rater or measurement agreement but also could be due to the lack of variability among the study sample, small number of participants and small number of raters being tested.23 In addition, the actual salt intake of the population and its variability may be an important factor. For example, Kawasaki equation was created in a high-sodium intake population, and therefore might overestimate sodium intake in other populations.24 INTERSALT was done in multiple countries in Europe and the Americas. In a study in Brazil, the estimation of salt consumption (difference between the measured and calculated salt consumption <1 g/day) was adequate only when the consumption was between 9 and 12 g/day (Tanaka) and 12 and 18 g/day (Kawasaki).25
A meta-analysis found that compared with the 24-hour samples, values based on spot urine overestimated the intake at lower levels of consumption and underestimated the intake at higher levels of consumption.26 A systematic review by Ji et al. reported a wide range of correlation coefficients between 24-hour urine sodium and other methods ranging between 0.94 and 0.17, with lower value corresponding to our coefficients.12 None of the systematic reviews or meta-analyses have been able to categorically identify one approach to be superior over the other. Another aspect to keep in mind is the need for measurement of potassium. While it may not be critical for sodium level estimation, potassium intake may increase urinary sodium excretion and can blunt the impact of sodium intake on blood pressure.27 It has been recommended that promoting the intake of potassium-rich foods along with programmes focused on sodium reduction may be more useful to improve heart health in India.28
Our study corroborates the findings of the only earlier study available from India20 in terms of low ICCs for all the three methods, with Kawasaki equation having the highest mean bias and overestimating the mean salt intake. INTERSALT and Tanaka performed similar as well in that study. The estimate of salt intake from Delhi (only site common to both the studies) was similar (8.6; 7.7–9.5 v. 8.0; 7.2–8.9). The other site of Andhra Pradesh in the study by Petersen et al.20 was in East India and neighbours the state of Odisha (Bhubaneswar site) and reported similar higher salt intake values. A study in South Africa also reported poor performance of all the three equations.29 The poor performance could be due to reasons such as physiological (body weight and gender differences), climatic (temperature and humidity) as well as operational (compliance to 24-hour collection and fasting instructions and use of morning sample). The low reported correlation in sodium levels between those estimated by the equations and measured 24-hour urine sample in our study can be due to both methodological and biological reasons.30 There are undoubtedly major challenges in standardized collection and measurement of urine samples as well as anthropometric measurements in field conditions and assessment methods of creatinine and sodium estimation in laboratories. Creatinine and BMI are the most important variables in the equations to estimate sodium, and they assume that the relationship between the three is constant across populations. However, studies have shown that it is not true. Creatinine levels are determined by muscular mass of a person, and we know that in the South Asian body type, BMI does not correlate well with the body fat or muscle mass.31 Similar hypotheses have been given for poor performance in South African populations.32
The strengths of our study were that it was sufficiently powered and covered individuals from different regions and demographic subgroups, and testing was done in a laboratory that met international quality standards. The limitations of the study were the operational challenges in collecting 24-hour urine samples, loss of study participants due to improper collection and transfer of samples. Also, the study did not aim at estimating population-level salt intake and therefore, did not follow a random sampling procedure for patient selection.
Conclusion
Only Tanaka equation provided acceptable values of 24-hour salt intake at the population level. INTERSALT equation (without potassium) is recommended by the WHO for enabling international comparisons. Using the same equation enables monitoring of salt intake over time. However, the poor performance of the equations highlights the need to understand the reasons and develop better methods for the measurement of sodium intake at the population level.
ACKNOWLEDGEMENTS
We acknowledge the help received from Dr Baridalyne Nogkynrih, Dr Kapil Yadav, Dr C.S. Pandav, Dr Vikas Bhatia, Late Dr Ramesh Chauhan, Bhoomika Bajaj Bhalla, Vaitheeshwaran Kulothungan and others for providing technical, logistical and field support for data collection and analysis. The study was funded by the WHO India Country Office under agreement 2016/671177–0.
Conflicts of interest
None declared
References
- Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet. 2016;388:1659-724.
- [Google Scholar]
- Salt reduction lowers cardiovascular risk: Meta-analysis of outcome trials. Lancet. 2011;378:380-2.
- [Google Scholar]
- Noncommunicable Diseases Global Monitoring Framework: Indicator Definitions and Specifications. 2014. Geneva: WHO; Available at www.who.int/nmh/ncd-tools/indicators/GMF_Indicator_Definitions_Version_NOV2014.pdf?ua=1 (accessed on 20 Jul 2018)
- [Google Scholar]
- World Health Organization The SHAKE Technical Package for Salt Reduction. 2016. Available at https://apps.who.int/iris/bitstream/handle/10665/250135/9789241511346eng.pdf;jsessionid=9CCA8A7B9A3617878EAB071AAD833AAF?sequence=1 (accessed on 10 Mar 2019)
- [Google Scholar]
- National approaches to monitoring population salt intake: A trade-off between accuracy and practicality? PLoS One. 2012;7:e46727.
- [Google Scholar]
- INTERSALT Co-Operative Research Group. Estimating 24-h urinary sodium excretion from casual urinary sodium concentrations in western populations: The INTERSALT study. Am J Epidemiol. 2013;177:1180-92.
- [Google Scholar]
- A simple method to estimate populational 24-h urinary sodium and potassium excretion using a casual urine specimen. J Hum Hypertens. 2002;16:97-103.
- [Google Scholar]
- A simple method for estimating 24 h urinary sodium and potassium excretion from second morning voiding urine specimen in adults. Clin Exp Pharmacol Physiol. 1993;20:7-14.
- [Google Scholar]
- Monitoring Population Salt Intake as Part of WHO STEPS. Report of a Meeting in Sydney 2013 December 2-4
- [Google Scholar]
- Expert Meeting on Population Sodium Reduction Strategies for Prevention and Control of Noncommunicable Diseases in the South-East Asia Region. In: Report of the Regional Meeting. New Delhi, India: WHO; 2012. SEA-NCD-88
- [Google Scholar]
- Use of urine biomarkers to assess sodium intake: Challenges and opportunities. Annu Rev Nutr. 2015;35:349-87.
- [Google Scholar]
- Systematic review of studies comparing 24-hour and spot urine collections for estimating population salt intake. Rev Panam Salud Publica. 2012;32:307-15.
- [Google Scholar]
- Urinary excretion of sodium, potassium, and chloride, but not iodine, varies by timing of collection in a 24-hour calibration study. J Nutr. 2013;143:1276-82.
- [Google Scholar]
- Validation and assessment of three methods to estimate 24-h urinary sodium excretion from spot urine samples in Chinese adults. PLoS One. 2016;11:e0149655.
- [Google Scholar]
- Estimation of populational 24-h urinary sodium and potassium excretion from spot urine samples: Evaluation of four formulas in a large national representative population. J Hypertens. 2017;35:477-86.
- [Google Scholar]
- Validation study of the Tanaka and Kawasaki equations to estimate the daily sodium excretion by a spot urine sample. Rev Bras Epidemiol. 2015;18(Suppl 2):224-37.
- [Google Scholar]
- Validation and comparison of three formulae to estimate sodium and potassium excretion from a single morning fasting urine compared to 24-h measures in 11 countries. J Hypertens. 2014;32:1005-14.
- [Google Scholar]
- Measuring population sodium intake: A review of methods. Nutrients. 2014;6:4651-62.
- [Google Scholar]
- Government of India. National Action Plan and Monitoring Framework for Prevention and Control of Non-Communicable Diseases in India. Available at https://mohfw.gov.in/sites/default/files/9967643039National_Action_PlanandMonitoring_Framework.pdf (accessed on 20 Jul 2018)
- [Google Scholar]
- Estimating population salt intake in India using spot urine samples. J Hypertens. 2017;35:2207-13.
- [Google Scholar]
- Method agreement analysis: A review of correct methodology. Theriogenology. 2010;73:1167-79.
- [Google Scholar]
- A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med. 2016;15:155-63.
- [Google Scholar]
- Estimating dietary sodium intake using spot urine samples: Correlation and bias. J Hypertens. 2017;35:466-7.
- [Google Scholar]
- Correlation between sodium and potassium excretion in 24-and 12-h urine samples. Braz J Med Biol Res. 2012;45:799-805.
- [Google Scholar]
- Mean population salt intake estimated from 24-h urine samples and spot urine samples: A systematic review and meta-analysis. Int J Epidemiol. 2016;45:239-50.
- [Google Scholar]
- Dietary reference intake for water, potassium, sodium, chloride, and sulfate. Washington, DC: National Acadamy Press;
- [Google Scholar]
- Potassium intake in India: Opportunity for mitigating risks of high-sodium diets. Am J Prev Med. 2020;58:302-12.
- [Google Scholar]
- Prediction of 24-hour sodium excretion from spot urine samples in South African adults: A comparison of four equations. J Hum Hypertens. 2020;34:24-33.
- [Google Scholar]
- Validation and comparison of three formulae to estimate sodium and potassium excretion from a single-morning fasting urine compared to 24-h measures in 11 countries. J Hypertension. 2014;32:2499-500.
- [Google Scholar]
- The thin-fat phenotype and global metabolic disease risk. Curr Opin Clin Nutr Metab Care. 2011;14:542-7.
- [Google Scholar]
- Monitoring the South African population’s salt intake: Spot urine v 24 h urine. Public Health Nutr. 2018;21:480-8.
- [Google Scholar]




