Translate this page into:
Non-operative treatment for low back pain: A review of evidence and recommendations
To cite: Garg B, George J, Mehta N. Non-operative treatment in low back pain: A review of current evidence and treatment recommendations. Natl Med J India 2022;35:19–27.
Abstract
Background
Low back pain (LBP) is a healthcare problem with high global prevalence, with non-operative management being the first line of treatment in the majority of patients. This literature review summarizes the current evidence for various modalities of non-operative treatment for LBP.
Methods
We did a literature search to elicit high-quality evidence for non-operative treatment modalities for LBP, including Cochrane Database reviews and systematic reviews or meta-analysis of randomized controlled trials. Only when these were not available for a particular treatment modality, other level 1 studies were included. The quality of evidence was categorized in accordance with the Grading of Recommendations, Assessment, Development and Evaluations (GRADE) method—a globally adopted tool for grading the quality of evidence and making treatment recommendations.
Results
The treatment modalities that were reviewed included: general measures, medications/pharmacotherapy, exercises, electromagnetic therapies, alternative treatment modalities and interventional therapies. We found that high-quality evidence is lacking for most non-operative treatment modalities for LBP. The majority of interventions have small benefits or are similar to placebo.
Conclusion
The current evidence for non-operative treatment modalities for LBP is insufficient to draw conclusions or make recommendations to clinicians. High-quality trials are required before widespread use of any treatment modality. Considering that non-operative treatment is usually the first line of therapy for most patients with LBP, it deserves to be the focus of future research in spinal disorders.
INTRODUCTION
Low back pain (LBP) is a common condition with about 80% of the population experiencing at least a single episode during their lifetime.1 For most practical purposes, LBP can be categorized into three types: (i) non-specific LBP; (ii) LBP with radicular symptoms; and (iii) LBP consequent to serious underlying pathologies (such as fracture, infection and malignancy).2 Non-specific LBP, the most common form, often has a chronic course with more than one-third of the patients being symptomatic beyond a year of its onset.3 Patients with chronic LBP tend to have a waxing and waning course with multiple recurrences, making it the second most common cause of disability in adults in the USA.4
Despite its varied presentation, the first-line treatment of LBP is centred on non-operative measures in the majority of patients. We discuss non-operative therapies and the supporting evidence. Wherever possible, we have included meta-analysis or systematic reviews of randomized controlled trials (RCTs). For interventions where a meta-analysis or systematic review was not available, level 1 studies or the highest-level studies were included. The quality of evidence was categorized when possible using the Grading of Recommendations, Assessment, Development and Evaluations (GRADE) method as high (confident that the true effect lies close to that of the estimate of the effect), moderate (moderately confident that the true effect is likely to be close to the estimate of the effect, but further research may change the effect), low (confidence in the effect estimate is limited and further research is likely to change the estimate of the effect) or very low (very little confidence in the effect estimate and the estimate is very uncertain).5
REVIEW OF TREATMENT METHODS
General measures
These typically home-based treatment modalities can be tried without a formal prescription or advice. They can be considered as a part of lifestyle measures to cope with LBP. Table I summarizes the current evidence for each of these measures whereas a detailed commentary follows.
| Modality | Summary of evidence | Quality of evidence* |
|---|---|---|
| Rest | Better with staying active versus rest in acute LBP | Moderate |
| No effects in sciatica | Moderate | |
| Similar benefits for staying active versus exercise/physiotherapy in acute LBP | Low | |
| Education | Better outcomes with intensive education in acute/sub-acute LBP (but not with less intensive education) | Strong |
| No benefit for education alone in chronic LBP | Low | |
| Better outcomes with addition of education to physiotherapy in chronic LBP | Moderate | |
| Heat therapy | Small short-term benefit in acute/sub-acute LBP | Moderate |
| Insufficient evidence for chronic LBP | – | |
| Cold therapy | Insufficient evidence | – |
| Lumbar support | No benefit in prevention or treatment of LBP | Moderate |
| Insoles | No benefit in prevention of LBP | Strong |
| Insufficient evidence for treatment of LBP | – |
Rest
Conventionally, rest was an important part of treatment for acute LBP. Later studies revealed that prolonged inactivity is potentially harmful to patients as it may lead to worsening of many body functions.6 Dahm et al. conducted a systematic review including 10 RCTs and found moderate evidence (including two trials) that patients with acute LBP may experience small benefits in functional improvement and pain relief from advice to stay active as opposed to advice for bed rest.7 However, there was little or no difference between the two approaches in patients with sciatica.7
Education
Education helps patients in adjusting with their back pain by providing information, advice and behaviour modification techniques. Providing information prevents unnecessary use of healthcare resources, enhances self-care and reduces unwarranted concerns about serious outcomes. Patient education is difficult, requires substantial effort and patience from the healthcare provider, and motivation and cooperation from the patient.8 Different types of patient education are used in clinical practice––oral or written, individual or to a group of patients, as a separate intervention or as part of other interventions. Engers et al.9 reviewed 24 RCTs investigating patient education in LBP and found strong evidence that intensive patient education is effective for patients with acute or sub-acute LBP, though less intensive education had no effect. In a meta-analysis by Wood and Hendrick,10 the authors found moderate-quality evidence that patient education was beneficial for pain and disability in chronic LBP.
Heat and cold therapy
Similar to other painful conditions, both heat and cold therapies have been used in LBP. Heat therapy works by increasing the blood circulation, which can theoretically reduce the inflammatory products in the heated tissues. Cold therapy, in contrast, is recommended for acute injuries, where it reduces the blood flow, preventing inflow of inflammatory products responsible for pain and oedema. A systematic review including nine clinical trials found moderate evidence (based on four trials using heat wrap or heated blanket) that there was a small, short-term reduction in pain and disability with heat therapy in patients with acute and sub-acute LBP. However, the evidence for cold therapy in LBP, and heat therapy in chronic LBP was limited, and no conclusions could be drawn.11 In another RCT, the authors did not find a difference between heat or cold therapy in patients with back and neck pain, suggesting that both might have comparable effects.12
Lumbar support
Lumbar supports or braces are used in the management of LBP both as a preventive or precautionary measure and as a treatment method (Fig. 1). Although the mechanism of lumbar supports is unclear, it is hypothesized that the lumbar braces correct deformity, limit spinal motion, provide additional stability and redistribute forces on the spine.13 In a Cochrane review of seven preventive RCTs (14 437 people) and eight treatment RCTs (1361 people), the authors found moderate evidence that when compared to no intervention, lumbar supports were not more effective in preventing or achieving short-term pain reduction in patients with acute or chronic LBP.14
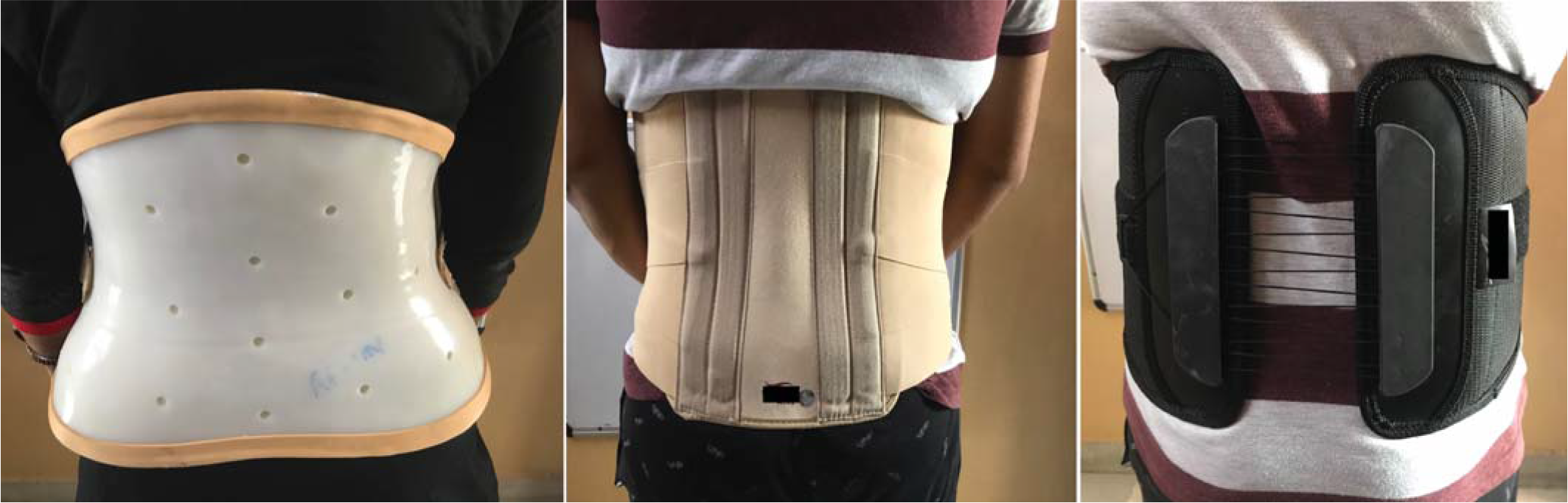
- Various types of lumbar supports in common use
Insoles
Difference in the length of the two limbs can cause imbalance of spine in the coronal plane, leading to uneven distribution of forces at the spine, which might contribute to early degeneration and back pain, making the case for use of shoe lifts in such patients. However, Brady et al. in their narrative review did not find a definite association in 12 studies that attempted to find out the link between limb length discrepancy (LLD) and LBP.15 Even in patients without clinically significant LLD, it is hypothesized that shoe insoles might be beneficial in the management of back pain by aiding shock absorption, preventing excessive movements of foot and improving balance. In a systematic review, strong evidence (based on three RCTs) was found that insoles are not effective in preventing LBP.16
MEDICATIONS
Many patients with acute LBP tend to have resolution of symptoms with time, and the treatment is aimed at controlling the pain during the episode. Patients are offered a variety of medications to control their symptoms either as a single agent or in combination. Table II summarizes the current evidence for each class of medication whereas a detailed commentary follows.
| Medication | Summary of evidence | Quality of evidence* |
|---|---|---|
| Paracetamol | No benefit in acute LBP | High |
| Inadequate evidence for chronic LBP | – | |
| NSAIDs | Slight benefit in acute LBP | Moderate to high |
| Slight benefit in chronic LBP | Low | |
| No benefit in sciatica | Very low to low | |
| No difference between NSAIDs | High | |
| Opioids | Better pain relief in chronic LBP | Low |
| Inadequate evidence for acute LBP | – | |
| Muscle relaxants | Short-term benefit in acute LBP | High |
| Insufficient evidence for chronic LBP, though benzodiazepines are effective | – | |
| Antidepressants | No clear benefit in chronic LBP, though might be useful in subset of patient (e.g. chronic pain syndromes, associated depression) | Moderate |
| Inadequate evidence for acute LBP | – | |
| Gabapentinoids | Ineffective in LBP and radicular pain | Moderate to high |
Paracetamol
Paracetamol (acetaminophen), one of the most commonly used medications worldwide, has antipyretic and analgesic properties. The usual recommended dose in adults is 325–1000 mg every 4 or 6 hours, not exceeding 4000 mg/day.17 Paracetamol is the initially preferred medication for many painful indications including LBP due to its low-risk profile. It is proposed that paracetamol acts by inhibition of cyclooxygenase (COX) and thus synthesis of prostaglandins.17 In a systematic review by Saragiotto et al., there was high-quality evidence that no difference existed between paracetamol (4000 mg/day) and placebo for acute LBP in terms of pain, function and adverse events.18 RCTs comparing paracetamol with placebo for patients with sub-acute or chronic LBP are lacking.
Non-steroidal anti-inflammatory drugs
Non-steroidal anti-inflammatory drugs (NSAIDs) are the most frequently prescribed class of medications worldwide for LBP. NSAIDs inhibit the activity of COX enzymes that are involved in the synthesis of a number of inflammatory mediators. Due to lower gastrointestinal side-effects, selective COX-2 inhibitors have become popular although there are concerns over their cardiovascular toxicity.19 In a meta-analysis of RCTs, high-quality evidence was found for NSAIDs being more effective than placebo in acute LBP though the effects were small. The authors also found moderate evidence that NSAIDs were not more effective than paracetamol for acute LBP with paracetamol having lower side-effects. In addition, there was strong evidence that various types of NSAIDs (including selective COX-2 inhibitors) are just as effective for acute LBP.20 In another systematic review, Enthoven et al. found that 6 of 13 RCTs showed that NSAIDs are more effective than placebo in reducing pain and disability in chronic LBP.21 However, the magnitude of the effects was small, while the level of evidence was low. In a systematic review of RCTs, Rasmussen-Barr et al. found NSAIDs not to be effective in reducing pain in sciatica.22
Opioids
Opioids have strong analgesic properties and act on opioid receptors in the central and peripheral nervous system. Primarily used for treating pain from malignancy, many practitioners are hesitant to use them in LBP due to the risk of drug dependence and abuse. Elderly patients with moderate-to-severe LBP who are less likely to indulge in drug abuse may be considered to be candidates for opioids, particularly because one might want to avoid prescribing NSAIDs to such patients.23 In review of 15 RCTs, Chaparro et al. found low-to-moderate-quality evidence for short-term efficacy of opioids to treat chronic LBP compared to placebo.24 In another meta-analysis, Petzke et al. compared studies with both a traditional RCT design and an enriched enrolment randomized withdrawal (EERW) design. In those with an EERW design, Petzke et al. noted that there was very low-to-low-quality evidence that there was clinically relevant pain relief with opioids compared to placebo in chronic LBP.25
Muscle relaxants
Painful spasm of paraspinal muscles is considered to be a protective mechanism in patients with LBP to guard the inflamed/ unstable vertebral elements. Broadly, muscle relaxants can be divided into two types: antispasmodic and antispasticity drugs. Antispasticity agents such as baclofen and dantrolene are used in improving muscle hypertonicity and involuntary jerks in spastic conditions such as multiple sclerosis, cerebral palsy and spinal cord injuries. On the other hand, antispasmodic agents such as cyclobenzaprine are better suited to treat musculoskeletal conditions such as LBP. Antispasmodics may be sub-classified into benzodiazepines and non-benzodiaze-pines. Benzodiazepines (e.g. diazepam, tetrazepam) also have anxiolytic, sedative and anticonvulsant effects in addition to skeletal muscle relaxation. Cyclobenzaprine, carisoprodol, metaxalone and chlorzoxazone are some commonly used nonbenzodiazepines antispasmodic agents in LBP.26 In a meta-analysis of 30 RCTs assessing the efficacy of muscle relaxants in LBP, strong evidence was found in favour of muscle relaxants being more effective than placebo for short-term relief of acute LBP; however, there is a need for caution in view of adverse effects.27 The evidence for use of muscle relaxants in chronic LBP is less convincing.
Antidepressants
Many patients with chronic LBP often have associated depression and other somatic symptoms. The analgesic, sedating and antianxiety properties of this class of medications help in the improvement of pain tolerance and quality of life in such patients.28 In a systematic review of 10 RCTs, no difference was found in pain relief between anti-depressant and placebo treatments.29 However, an earlier systematic review by Salerno et al. concluded that anti-depressants are more effective in chronic LBP than placebo.30 This is probably because Salerno et al. included some studies that were not adequately randomized and had patients with other pain syndromes in addition to LBP. The evidence for use of antidepressants in acute LBP is unclear.
Gabapentinoids
Many patients with LBP have symptoms and signs of nerve irritation ranging from leg pain to dermatomal paraesthesia or even weakness in rare cases. Gabapentinoids (such as gabapentin and pregabalin) have anticonvulsant and analgesic properties and work by limiting neuronal excitation and enhancing inhibition.31 Their effectiveness has been well proven previously in neuropathic pain conditions such as diabetic neuropathy and phantom limb pain.32 In a meta-analysis by Enke et al., nine clinical trials comparing topiramate, gabapentin or pregabalin to placebo in 859 patients were included. They found moderate-to-high-quality evidence that gabapentinoids are ineffective in the treatment of LBP or for lumbar radicular pain.33 A majority of these studies included patients with chronic LBP, though Mathieson et al. included patients with both acute and chronic symptoms and reached similar conclusions.34
EXERCISE
By improving the muscle imbalance around the lower back and pelvis, exercise therapies ameliorate the lumbar stability and reduce the stress on disc and posterior vertebral elements. Different exercise regimens include one or more of the following components: aerobic exercises, strengthening of lumbar extensor muscles, strengthening of abdominals and flexor group of muscles and exercises to improve stability of the pelvis. In a meta-analysis, Hayden et al. included 61 RCTs and found that in acute LBP, there is moderate evidence that exercises are not more effective than other conservative treatments. In sub-acute LBP, there was moderate evidence of effectiveness of a graded activity exercise programme in reducing absenteeism at work. There was strong evidence that exercise was at least as effective as other conservative treatment modalities with overall better outcomes in chronic LBP—although the effects were very small.35 Choi et al. performed a meta-analysis and found moderate-quality evidence that exercises reduced the rate of recurrence of LBP at one year.36 Although different regimens have been proposed, there is limited evidence to support that one form of exercise is superior to other. Table III summarizes the current evidence for different forms of exercise for LBP, whereas a detailed commentary follows.
| Modality | Summary of evidence | Quality of evidence* |
|---|---|---|
| All exercises | No effect in acute pain | Moderate |
| Some benefit with graded programme in sub-acute pain | Moderate | |
| Small benefits in chronic pain | Moderate to high | |
| Prevents recurrence | Moderate | |
| Muscle energy technique | No effect | Low |
| Motor control exercises | Clinically important benefit in chronic LBP | Low to moderate |
| No benefit in acute LBP | Very low | |
| Yoga | Small benefit in chronic LBP compared with no exercise | Low |
| Similar to other exercises | Very low | |
| Pilates | Moderate benefit in LBP compared to minimal intervention | Low to moderate |
| Similar to other exercises | Very low | |
| Multidisciplinary rehabilitation | Better outcomes in sub-acute LBP compared to usual treatment | Low |
| Moderate benefit in chronic LBP compared to usual treatment | Moderate |
Muscle energy technique (MET)
In this technique, the operator identifies one particular muscle or muscle group, and the patient actively contracts the muscle in a specific direction while the operator applies a steady counterforce. It is believed to work by reducing localized oedema and inflammation in tissues and improve tolerance due to pain modulation.37 One notable limitation with MET is that the technique is operator-dependent. In a meta-analysis of 12 RCTs, the authors found low-quality evidence that MET was not effective for patients with LBP.38
Motor control exercises (MCE) or lumbar stabilization exercises
This form of exercise targets the muscles around the lumbar spine to restore their control and coordination. The programmes usually start with simple static exercises and progress into more complex and dynamic movements involving the activation of various deep trunk muscles. The MCE is a dynamic intervention that needs constant adjustment based on the patients’ performance/ response.39 However, many exercise regimens include only the training of deep muscles in the intervention and do not take into consideration the principles of motor control and progressive learning. Therefore, studies assessing the effectiveness of MCE generally include all forms of lumbar stabilization exercises. In a systematic review of 29 clinical trials for chronic LBP, the authors found low-to-moderate-quality evidence that compared to minimal intervention, MCE had a clinically important beneficial effect.40 In another systematic review, the authors found very low evidence that MCE was not effective in patients with acute LBP.41
Yoga
Yoga is a set of physical, mental and spiritual practices originating from ancient India, which has become popular around the world. It is being increasingly used for therapeutic purposes such as managing chronic pain and disability associated with musculo-skeletal conditions. In a 2002 National Health Interview Survey (Alternative Medicine Supplement survey), among 10 million Americans adults using yoga, about 10% used yoga for musculoskeletal conditions.42 It is assumed that yoga improves flexibility and strength of the trunk muscles as a result of its various physical poses (Fig. 2). In addition, the breathing and meditation exercises are thought to improve the overall body awareness and mental fitness improving pain tolerance. In a systematic review of 12 RCTs (1080 participants), the authors found low evidence that yoga compared to non-exercise controls resulted in small improvements in pain intensity in patients with chronic LBP. There was also low-to-moderate evidence that yoga improved function.43

- Photographs of yoga Asana(s) for improving back pain, increasing general awareness and mental function
Pilates
Developed by Joseph Hubertus Pilates, this exercise form works on improving overall body awareness and posture. Pilates exercises are based on the following traditional principles: centring (tightening the muscular centre), concentration (cognitive attention to perform the exercise), control (management of posture and control), precision (accuracy of exercise technique), flow (smooth transition of movements within the exercise sequence) and breathing (in coordination with the movements).44 Pilates exercises are usually prescribed by certified instructors in individual or group sessions. In a review of 10 RCTs with chronic LBP, Yamato et al. found lowto-moderate evidence that pilates reduced pain (medium effect size) compared to minimal intervention.45
Multidisciplinary rehabilitation programmes
Despite the large number of treatment options, the management of LBP by any single modality is often not very rewarding. It has been acknowledged that chronic LBP is not merely a physical disorder, but a combination of physical, social and psychological dysfunctions. Multidisciplinary rehabilitation programmes are delivered by a team of healthcare workers—these are characterized by a physical component (usually some form of exercise) along with a psychological, social or occupational component. In a meta-analysis of nine RCTs, low-to-very low-quality evidence was found for multidisciplinary approach performing better than usual care in sub-acute LBP.46 In a meta-analysis of 41 RCTs for chronic LBP, there was moderate-quality evidence that multidisciplinary treatment improved pain and function to a larger extent than usual care or treatment which targeted only physical factors.47
ALTERNATIVE TREATMENT MODALITIES
These treatment methods are not typically prescribed by allopathic doctors and are usually performed by various practitioners trained in these modalities. Table IV summarizes the current evidence for various alternative treatment modalities for LBP as we discuss some of them in this section.
| Modality | Summary of evidence | Quality of evidence* |
|---|---|---|
| Spinal manipulative therapies | Small benefit, but not clinically significant, in chronic LBP | High |
| No benefit in acute LBP | Very low to low | |
| Chiropractic intervention | No benefit in chronic LBP | Low |
| Small benefit, but not clinically significant, in chronic LBP | Low | |
| Acupuncture | Short-term benefit in chronic LBP | Low |
| Insufficient evidence in acute LBP | – | |
| Massage | Massage better than controls in acute LBP | Very low |
| Short-term benefit in chronic LBP (similar to small benefit in the long term) | Very low to low |
Spinal manipulative therapy typically uses high-velocity, low-amplitude thrusts applied to the vertebral joints. The putative modes of action of spinal manipulative therapy can be explained by mechanical and neurophysiological models. The mechanical model is based on the thought that there are vertebral subluxations or joint abnormalities, which when corrected can reduce the symptoms. The neurophysiological model suggests that spinal manipulation therapy affects the primary afferent neurons from paraspinal tissues, which alters the pain sensation.48 In a meta-analysis of 26 RCTs, Rubinstein et al. found high-quality evidence that there is a small statistically significant, but not a clinically relevant difference between spinal manipulation therapies and other interventions as far as reduction in pain and improvement in function in chronic LBP were concerned.49 In another meta-analysis, the same group of authors found low-to-very low-quality evidence suggesting no difference between spinal manipulation therapies and other interventions for acute LBP.50
In acupuncture, a form of therapy with Chinese origins, various acupuncture points described in traditional texts are stimulated by acupuncture needle insertions. Dry needling is a technique closely resembling acupuncture that is used in myofascial syndromes. Dry needling differs from trigger point injections in that a medication such as a steroid or local anaesthetic is injected in the latter case. The gate control theory of pain has been attributed as the underlying principle behind the effectiveness of acupuncture.51 In a systematic review of five RCTs evaluating the effectiveness of acupuncture or dry needling, Furlan et al.52 found that there is low-quality evidence of pain relief and improvement in function for acupuncture, compared to no treatment in chronic LBP with these effects predominantly only at short-term follow-up. The authors could not make firm conclusions with respect to acute LBP.
Therapeutic massage is another popular treatment for LBP— it is thought to release endorphins and raise the pain threshold based on the gate control theory.53 In a systematic review of the effectiveness of massage in LBP, Furlan et al.54 found low-quality evidence for massage being better than a sham therapy in acute LBP. For chronic LBP, the authors found that massage was better than inactive controls for pain and function in the short-term, but not in the long-term follow-up. The overall evidence was very low to low.
ELECTROMAGNETIC INTERVENTIONS
These treatment modalities are based on the delivery of an electromagnetic energy in the form of electrical current, heat or shock waves to deeper tissues. Most of these therapies are used in patients with chronic LBP who have not responded to other simple measures. Table V summarizes the current evidence for various electromagnetic interventions for LBP as we discuss some of them in this section.
| Modality | Summary of evidence | Quality of evidence* |
|---|---|---|
| TENS | Beneficial in chronic LBP | Low |
| No benefit in acute LBP | Very low | |
| Interferential therapy | Insufficient evidence (likely similar to placebo) | – |
| Ultrasound diathermy | No benefit | Low |
| Microwave diathermy | No benefit | Very low |
| Short-wave diathermy | No benefit | Very low |
| Low-level laser therapy | Small, clinically insignificant benefit | Moderate |
| High-level laser therapy | Likely beneficial | Very low |
Transcutaneous electrical nerve stimulation (TENS)
The TENS is commonly used as an adjunctive treatment modality in the management of LBP. It is safe, non-invasive and can be administered at clinic or at home with ease. Believed to act on the basis of the gate control theory of pain, it consists of electrodes placed over the skin which deliver electrical currents to stimulate the deep nerves involved in pain transmission. Inhibitory interneurons in the dorsal horn of the spinal cord are activated when large diameter primary sensory afferent fibres are stimulated, which then block the transmission of nociceptive signals from small diameter fibres.55 The two most common types of TENS are high-frequency or conventional TENS (frequency >80 Hz, pulse width <150 μs) and low-frequency or acupuncture-like TENS (frequency <10 Hz).56 In a systematic review including five RCTs comparing TENS with placebo, Khadilkar et al.57 found conflicting evidence regarding the effectiveness of TENS in chronic LBP. In a meta-analysis of two RCTs assessing the effectiveness of TENS in acute LBP, the authors did not find any reduction in pain.58
Interferential therapy (IFT)
IFT uses the principle of interference-mixing two waves of different frequencies to produce a combined wave of another frequency at a distance. In IFT, medium-frequency currents are delivered at the skin, which brings about an interferential current of a low frequency in the deep tissues (Fig. 3). In an RCT of 150 patients with non-specific chronic LBP, Facci et al.59 compared TENS and IFT with controls. They found that both TENS and IFT reduced pain in comparison to controls who did not receive any treatment.
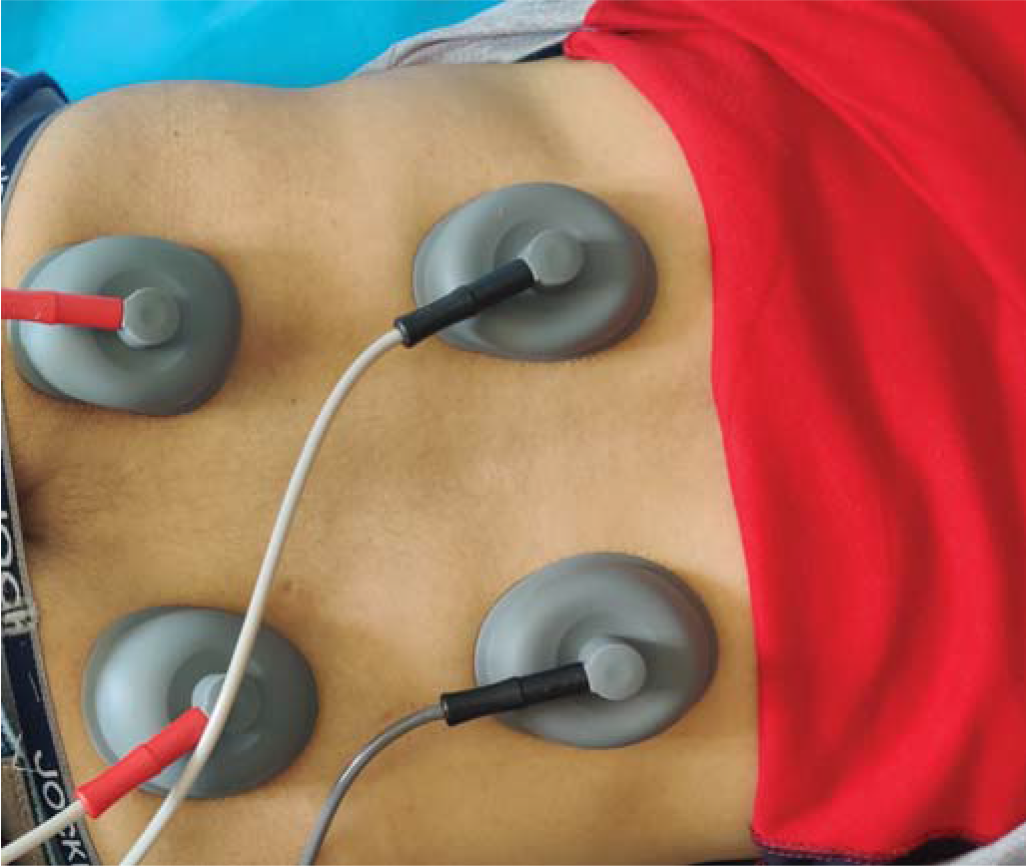
- Photograph of interferential therapy (IFT) being given to a patient with low back pain
Diathermy
Diathermy refers to the delivery of electromagnetic energy transcutaneously to heat up the underlying tissues, which can increase the blood flow to tissues. It works similar to superficial heat therapy applied on the skin, but at greater depths. Diathermy can be delivered using short wave, microwave or ultrasound. Short-wave diathermy uses high-frequency (short-wave length) electromagnetic waves to generate heat, while microwaves use electromagnetic waves in the microwave range. Short waves can penetrate much deeper tissues, while microwaves are limited to more superficial tissues. Ultrasound diathermy delivers energy to deep tissue sites through ultrasonic waves, which are similar to sound waves in properties. In addition to the thermal effects, therapeutic ultrasound is assumed to have mechanical effects characterized by the formation of microbubbles, which oscillate and produce a massage effect at the cellular level.60 In a review of seven RCTs, Ebadi et al.61 did not find any high-quality evidence to support the use of ultrasound for improving pain or quality of life in patients with non-specific chronic LBP. Although few small trials have suggested that both short-wave and microwave diathermy can be used as adjuncts in the management of LBP, there is lack of enough evidence to support its use.
Laser therapy
In laser therapy, tissues are irradiated by lasers or light of a single wavelength. Conventionally, low-level laser therapy is used for pain disorders and they act by non-thermal or photochemical effects on cells such as stimulating fibroblast function and accelerating connective tissue repair. Low-level laser may have anti-inflammatory effects due to its action in reducing prostaglandin synthesis.62 In a systematic review of seven RCTs, low-level laser therapy was found to reduce pain and disability in patients with sub-acute or chronic LBP, although treatment effects were small.63
INTERVENTIONAL THERAPIES
Interventional therapies are usually pursued when physical therapy and analgesics fail to relieve the patient’s symptoms. Table VI summarizes the current evidence for various interventional therapies for LBP as we discuss some of them in this section.
| Modality | Summary of evidence | Quality of evidence* |
|---|---|---|
| Radiofrequency ablation | Better than placebo in facet joint pain | Moderate |
| No benefit in discogenic pain, sacroiliac joint (SIJ) pain and radicular pain | Very low to low | |
| Prolotherapy | No benefit | Moderate |
| Epidural injections | No benefit | Moderate |
| Transforaminal better than other approaches | Very low | |
| Intra-articular injections | No benefit for facet joint injection | Moderate |
| SIJ injection has no therapeutic value, but might have diagnostic value | Low |
Radiofrequency ablation (RFA)
In RFA, electric current is passed through an electrode placed in the vicinity of the nerve responsible for transmission of pain. An alternating current with a frequency of 250–500 kHz is generally used, which generates heat and results in coagulation of the indicted sensory nerves impeding the conduction of nociceptive impulses.64 RFA can be used to treat facet joint pain (targeting the medial branch of dorsal ramus), discogenic pain, sacroiliac joint (SIJ) pain and radicular pain (dorsal root ganglion block). A systematic review of 23 RCTs by Maas et al.65 included 12 studies examining facet joint pain, five studies on disc pain, two studies on SIJ pain, two studies on radicular chronic LBP and one study on chronic LBP. They found moderate-quality evidence that RFA may relieve facet joint pain and improve function better when compared with placebo over a short-term period. There was very low-to-low-quality evidence that RFA is better than steroid injection for facet joint pain. For patients with disc pain, SIJ pain and radicular pain, there was very low-to-low-quality evidence that RFA had no effect compared with placebo.
Prolotherapy
In prolotherapy, an irritant solution is injected into the painful ligaments or its insertion sites. This is believed to result in activation of inflammatory cells, leading to release of growth factors and collagen deposition, thereby strengthening the ligaments, and improve the joint stability, leading to a reduction in pain.66 There are three major classes of solutions used in prolotherapy: irritants, chemotactic agents and osmotics. Yelland et al.67 identified five high-quality RCTs with a total of 366 participants that compared prolotherapy with other injections or treatments. The authors found that while prolotherapy is not an effective treatment for chronic LBP when used alone, it may improve symptoms when used in combination with other treatment modalities.
Epidural injections
Injection of steroids into the epidural space has been practised for decades in patients with LBP with an aim to reduce the inflammation in irritated nerve roots. These injections can be performed blindly using anatomical landmarks or using fluoroscopic or ultrasound guidance for better accuracy. Injections can be given using interlaminar, caudal or transforaminal approach. In a systematic review by Staal et al.,68 there was moderate evidence that epidural injections did not have a significant effect on pain. They also found that the effect of epidural steroid injections is not significantly different from NSAIDs. Multiple studies have also compared different approaches of epidural injections. However, in a systematic review by Liu et al.,69 there was no clinical or statistical difference between transforaminal and caudal approaches.
Intra-articular injections
These injections (Fig. 4) of steroids and local anaesthetics into the facet and SIJ have been tried. As both facet and SIJ are small joints, intra-articular placement of the needle may not be successful despite image guidance, leading to periarticular or partial intra-articular injections. Both intra-articular and periarticular injections are believed to yield similar results, and studies often include both the methods. In a Cochrane review by Staal et al.,68 there was moderate evidence that corticosteroid facet joint injections are not significantly different from placebo injections. This was supported by another review that showed limited utility of facet joint injections, though injection with local anaesthetic was found to have diagnostic utility.70 Hansen et al. systematically reviewed studies evaluating SIJ injections and found that the evidence for intra-articular steroid injections is poor for short- and long-term relief.71 However, its diagnostic utility was found to be good by Simopoulos et al.72
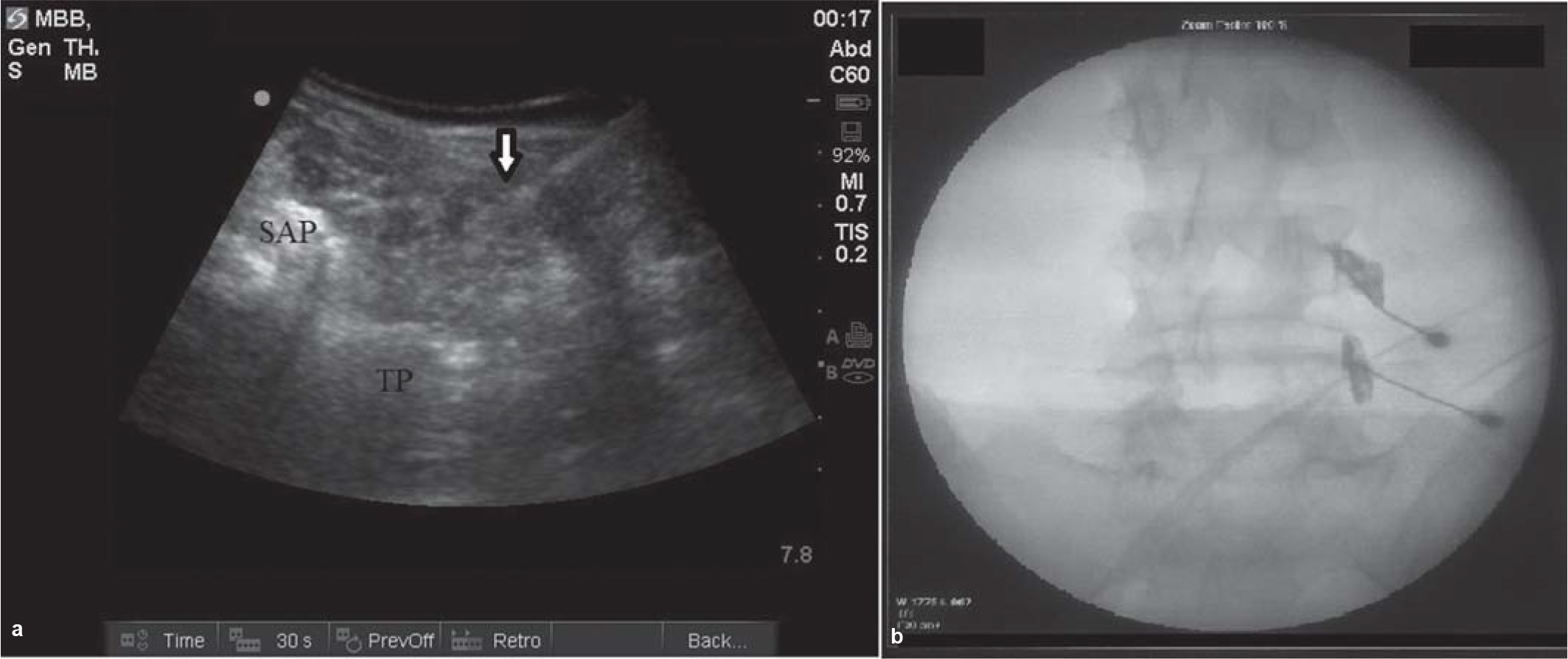
- Image-guided lumbar facet (medial bundle branch) block: (a) given under ultrasound guidance; (b) given under fluoroscopy guidance, showing spread of dye at two levels TP transverse process SAP superior articular process white arrow = needle
CONCLUSION
In this review, we found that very few interventions are supported by high-quality evidence. A majority of the non-operative measures are similar to placebo or have low or inadequate evidence in LBP. Exercise and simple analgesics appear to be the most effective interventions in LBP with strong supporting evidence. Despite the lack of strong evidence, many interventions are still being widely prescribed. As ineffective interventions contribute to the economic burden, it is important to formulate guidelines to manage LBP with interventions that have proven benefit. Moreover, well-designed trials are required to support current treatment modalities for LBP, which have not been adequately studied.
Conflicts of interest
None declared
References
- Predicting who develops chronic low back pain in primary care: A prospective study. BMJ. 1999;318:1662-7.
- [Google Scholar]
- Prevalence of disabilities and associated health conditions among adults––United States, 1999. MMWR Morb Mortal Wkly Rep. 2001;50:120.
- [Google Scholar]
- Bed rest: A potentially harmful treatment needing more careful evaluation. Lancet. 1999;354:1229-33.
- [Google Scholar]
- Advice to rest in bed versus advice to stay active for acute low-back pain and sciatica. Cochrane Database Syst Rev. :CD007612.
- [Google Scholar]
- The patient from receiver of information to informed decision-maker. Patient Educ Couns. 1998;34:89-102.
- [Google Scholar]
- Individual patient education for low back pain. Cochrane Database Syst Rev. 2008;2008(1):CD004057.
- [Google Scholar]
- A systematic review and meta-analysis of pain neuroscience education for chronic low back pain: Short-and long-term outcomes of pain and disability. Eur J Pain. 2019;23:234-49.
- [Google Scholar]
- Superficial heat or cold for low back pain. Cochrane Database Syst Rev. 2006;2006(1):CD004750.
- [Google Scholar]
- Heat or cold packs for neck and back strain: A randomized controlled trial of efficacy. Acad Emerg Med. 2010;17:484-9.
- [Google Scholar]
- Mechanisms of action of lumbar supports: A systematic review. Spine (Phila Pa 1976). 2000;25:2103-13.
- [Google Scholar]
- Lumbar supports for prevention and treatment of low back pain. Cochrane Database Syst Rev. 2008;2008(2):CD001823.
- [Google Scholar]
- Limb length inequality: Clinical implications for assessment and intervention. J Orthop Sports Phys Ther. 2003;33:221-34.
- [Google Scholar]
- Insoles for prevention and treatment of back pain. Cochrane Database Syst Rev. :CD005275.
- [Google Scholar]
- The modern pharmacology of paracetamol: Therapeutic actions, mechanism of action, metabolism, toxicity and recent pharmacological findings. Inflammopharmacology. 2013;21:201-32.
- [Google Scholar]
- Nonsteroidal antiinflammatory drugs, acetaminophen, and the risk of cardiovascular events. Circulation. 2006;113:1578-87.
- [Google Scholar]
- Non-steroidal anti-inflammatory drugs for low back pain. Cochrane Database Syst Rev. :CD000396.
- [Google Scholar]
- Non-steroidal anti-inflammatory drugs for chronic low back pain. Cochrane Database Syst Rev. 2016;2:CD012087.
- [Google Scholar]
- Non-steroidal anti-inflammatory drugs for sciatica. Cochrane Database Syst Rev. 2016;10:CD012382.
- [Google Scholar]
- What factors affect physicians’ decisions to prescribe opioids for chronic noncancer pain patients? Clin J Pain. 1997;13:330-6.
- [Google Scholar]
- Opioids compared with placebo or other treatments for chronic low back pain: An update of the Cochrane Review. Spine (Phila Pa 1976). 2014;39:556-63.
- [Google Scholar]
- Opioids for chronic low back pain. An updated systematic review and meta-analysis of efficacy, tolerability and safety in randomized placebo-controlled studies of at least four weeks double-blind duration. Eur J Pain. 2020;24:497-517.
- [Google Scholar]
- Muscle relaxants for nonspecific low back pain: A systematic review within the framework of the Cochrane Collaboration. Spine (Phila Pa 1976). 2003;28:1978-92.
- [Google Scholar]
- Antidepressants for non-specific low back pain. Cochrane Database Syst Rev. 2008;2008(1):CD001703.
- [Google Scholar]
- The effect of antidepressant treatment on chronic back pain: A meta-analysis. Arch Intern Med. 2002;162:19-24.
- [Google Scholar]
- The mechanisms of action of gabapentin and pregabalin. Curr Opin Pharmacol. 2006;6:108-13.
- [Google Scholar]
- Antiepileptic drugs for neuropathic pain and fibromyalgia––An overview of Cochrane reviews. Cochrane Database Syst Rev. 2013;2013(11):CD010567.
- [Google Scholar]
- Anticonvulsants in the treatment of low back pain and lumbar radicular pain: A systematic review and meta-analysis. CMAJ. 2018;190:E786-93.
- [Google Scholar]
- Trial of pregabalin for acute and chronic sciatica. N Engl J Med. 2017;376:1111-20.
- [Google Scholar]
- Exercise therapy for treatment of non-specific low back pain In: Cochrane Database Syst Rev. 2005. p. :CD000335.
- [Google Scholar]
- Exercises for prevention of recurrences of low-back pain. Cochrane Database Syst Rev. 2010;2010(1):CD006555.
- [Google Scholar]
- Muscle energy technique for chronic obstructive pulmonary disease: A systematic review. Chiropr Man Therap. 2019;27:37.
- [Google Scholar]
- Muscle energy technique for non-specific low-back pain. Cochrane Database Syst Rev. :CD009852.
- [Google Scholar]
- Motor control exercise for persistent, nonspecific low back pain: A systematic review. Phys Ther. 2009;89:9-25.
- [Google Scholar]
- Motor control exercise for chronic non-specific low-back pain. Cochrane Database Syst Rev. 2016;2016(1):CD012004.
- [Google Scholar]
- Motor control exercise for acute non-specific low back pain. Cochrane Database Syst Rev. 2016;2:CD012085.
- [Google Scholar]
- Prevalence and patterns of adult yoga use in the United States: Results of a national survey. Altern Ther Health Med. 2004;10:44-9.
- [Google Scholar]
- Yoga treatment for chronic non-specific low back pain. Cochrane Database Syst Rev. 2017;2017:1.
- [Google Scholar]
- Defining Pilates exercise: A systematic review. Complement Ther Med. 2012;20:253-62.
- [Google Scholar]
- Multidisciplinary biopsychosocial rehabilitation for subacute low back pain. Cochrane Database Syst Rev. 2017;6:CD002193.
- [Google Scholar]
- Multidisciplinary bio-psycho-social rehabilitation for chronic low-back pain. Cochrane Database Syst Rev. :CD000963.
- [Google Scholar]
- Spinal manipulative therapy for chronic low-back pain: An update of a Cochrane review. Spine (Phila Pa 1976). 2011;36:E825-46.
- [Google Scholar]
- Spinal manipulative therapy for acute low-back pain. Cochrane Database Syst Rev. 2012;2012(9):CD008880.
- [Google Scholar]
- Acupuncture and dry-needling for low back pain. Cochrane Database Syst Rev. :CD001351.
- [Google Scholar]
- The consistency of pulse frequencies and pulse patterns of transcutaneous electrical nerve stimulation (TENS) used by chronic pain patients. Pain. 1991;44:231-4.
- [Google Scholar]
- Transcutaneous electrical nerve stimulation (TENS) versus placebo for chronic low-back pain. Cochrane Database Syst Rev. 2008;2008(4):CD003008.
- [Google Scholar]
- Transcutaneous electric nerve stimulation (TENS) for acute low back pain: Systematic review. Scand J Pain. 2019;19:225-33.
- [Google Scholar]
- Effects of transcutaneous electrical nerve stimulation (TENS) and interferential currents (IFC) in patients with nonspecific chronic low back pain: Randomized clinical trial. Sao Paulo Med J. 2011;129:206-16.
- [Google Scholar]
- Therapeutic ultrasound for chronic low-back pain. Cochrane Database Syst Rev. :CD009169.
- [Google Scholar]
- The efficacy of laser therapy for musculoskeletal and skin disorders: A criteria-based meta-analysis of randomized clinical trials. Phys Ther. 1992;72:483-91.
- [Google Scholar]
- Low level laser therapy for nonspecific low-back pain. Cochrane Database Syst Rev. 2008;2008(2):CD005107.
- [Google Scholar]
- Electric and thermal field effects in tissue around radiofrequency electrodes. Pain Med. 2005;6:405-24.
- [Google Scholar]
- Radiofrequency denervation for chronic low back pain. Cochrane Database Syst Rev. 2015;2015(10):CD008572.
- [Google Scholar]
- Prolotherapy: A clinical review of its role in treating chronic musculoskeletal pain. PM R. 2011;3(6 Suppl 1):S78-S81.
- [Google Scholar]
- Prolotherapy injections for chronic low-back pain. Cochrane Database Syst Rev. :CD004059.
- [Google Scholar]
- Injection therapy for subacute and chronic lowback pain. Cochrane Database Syst Rev. 2008;2008(3):CD001824.
- [Google Scholar]
- The effectiveness of transforaminal versus caudal routes for epidural steroid injections in managing lumbosacral radicular pain: A systematic review and meta-analysis. Medicine (Baltimore). 2016;95:e3373.
- [Google Scholar]
- Systematic assessment of diagnostic accuracy and therapeutic utility of lumbar facet joint interventions. Pain Physician. 2009;12:437-60.
- [Google Scholar]
- A systematic evaluation of the therapeutic effectiveness of sacroiliac joint interventions. Pain Physician. 2012;15:E247-78.
- [Google Scholar]
- A systematic evaluation of prevalence and diagnostic accuracy of sacroiliac joint interventions. Pain Physician. 2012;15:E305-44.
- [Google Scholar]




