Translate this page into:
Renal outcomes among snake-envenomed patients with acute kidney injury in southern India
2 Department of General Medicine, Pondicherry Institute of Medical Science (PIMS), Puducherry, India
3 Department of Biostatistics, Christian Medical College, Vellore 632004, Tamil Nadu, India
Corresponding Author:
Tarun K George
Department of General Medicine, Christian Medical College, Vellore 632004, Tamil Nadu
India
tarunkg@gmail.com
| How to cite this article: George TK, Toms AG, Fenn BN, Kumar V, Kavitha R, Georgy JT, Abraham G, Zachariah A. Renal outcomes among snake-envenomed patients with acute kidney injury in southern India. Natl Med J India 2019;32:5-8 |
Abstract
Background. Acute kidney injury (AKI) is a common complication of snake envenomation. However, the long-term renal outcomes of such patients are not well defined. We aimed to determine the proportion of patients who developed AKI, characterize the presenting syndromes and ascertain the long-term resolution of AKI.Methods. We did a cohort study with prospective follow- up from two centres in southern India. All admitted patients >15 years of age with snake envenomation and serum creatinine ≥1.5 mg/dl over the past 10 years were identified through their discharge summaries. These patients were prospectively contacted, interviewed telephonically and requested to come for a hospital review.
Results. Of the 866 patients screened, 1 84 developed AKI (21.2%). Among these, 53% had combined renal, haematological and neurological manifestations; 33.6% required admission to the intensive care unit and 38% were dialysed. On follow-up of hospital records the creatinine of 49% of patients had normalized. Of those admitted, 36% were contacted and none had a known renal disease or were on dialysis. Among these, 16 patients came to the hospital for review and only 2 had an elevated creatinine. The total mortality was 1 4.
Conclusion. AKI is an important cause of morbidity with snake envenomation and a proportion will require dialysis. The mortality in our study was low and long-term renal outcomes were relatively good.
Introduction
Snake envenoming is an important yet neglected tropical disease. It has a rural predisposition and is associated with strong economic and health implications. Globally, the estimated snake envenoming is around 5.5 million annually[1] and, in India, it is estimated that about 45 900 deaths occur annually due to snake bite.[2] The ‘Indian Big four’ have been implicated in the majority of serious snake bites in India. They are the Viperidae—Daboia russelii (Western Russell’ s viper), Echis carinatus (Saw-scaled Viper), Elapidae— Naja naja (Spectacled Cobra) and Bungarus caeruleus (Common Krait).
The overall incidence of complications following snake bite envenoming varies from 30% to 40%.[3],[4] The common clinical effects of envenoming are local toxicity, lymphadenitis, haematological (coagulopathy and thrombocytopenia), renal injury and neurotoxic manifestations.[4]·[5] Acute kidney injury (AKI) is a common development, ranging from 13% to 25%.[4],[6],[7] Syndromespecies correlation studies in Sri Lanka have shown that the snake species commonly associated with AKI are Russell’s viper and Hump-nosed viper (Hypnale hypnale).[8],[9] Often, the culprit snake is not identified. WHO has described five clinical syndromes indicative of the snake responsible. Renal involvement is associated with the Russel’s viper and sea snakes.[10] In our region, vipers commonly responsible are Russel’ s and saw-scaled vipers. Patients bitten by these snakes also present with local and haematological features.
Despite the high incidence of AKI with snake envenoming, information about the long-term renal function in patients after discharge is limited. Studies from Kolkata and Sri Lanka have noted that chronic kidney disease (CKD) develops in 41% and 37%, respectively, ofthe snake bitten population following AKI.[11],[12] We have not encountered such a high rate of progression to CKD. Clinical and venom-based data suggest a geographical variation in the envenoming species and also in the nature of the venom.[13],[14] A review of geographical variation of snake bite syndromes showed that in southern India snake bites were predominantly haemotoxic with renal failure and in northern India they were predominantly neurotoxic. Russell’s viper bites were scarce in northern India.[15] Hence, a different profile of clinical features and outcomes may be observed in patients in our hospitals. We aimed to determine the incidence of AKI in our setting, characterize the presenting syndromes and ascertain the long-term renal outcomes in terms of resolution or progression to CKD.
Methods
Our cohort study had retrospective recruitment and prospective follow-up of patients with snake envenoming-induced AKI, who had been admitted to the departments of general medicine. Two centres involved in this study—Christian Medical College (CMC) Hospital, Vellore, Tamil Nadu, and Pondicherry Institute of Medical Sciences (PIMS), Puducherry—are tertiary academic institutions in southern India. Data were extracted from the discharge summaries. CMC had online hospital information and PIMS had physical inpatient hospital records. We screened data of patients who presented from January 2005 to December 2015. From the follow-up of online records and files, the serum creatinine at discharge was noted. The patients were then contacted and interviewed telephonically. Details of their present health complaints, comorbid conditions and any known kidney problem were determined. They were requested to come to the hospital for a clinical and laboratory review. The follow-up was done by three methods: through follow-up of computer-based creatinine values, telephonic interview and a hospital visit for check-up and tests.
The inclusion criteria were patients aged >15 years with an alleged history of snake bite admitted to medical wards with a serum creatinine of ≥1.5 mg/dl. Patients with pre-existing CKD were excluded.
We adopted the AKI definition from The Kidney Disease: Improving Global Outcomes (KDIGO) Clinical Practice Guidelines for AKI (2012).[16] We assumed that if the baseline creatinine was 1 mg/dl, a value of 1.5 mg/dl would reflect stage I severity of AKI. We described CKD with reference to the KDIGO 2013 guideline.[17] We assumed that a serum creatinine of ≤1.3 mg/dl would be normal with an estimated glomerular filtration rate of >60 ml/minute/1.73 m[2]. Hence, all such patients were considered to be normal during follow-up.
Results
Retrospective data
The number of patients screened was 866 (CMC 746 and PIMS 120). The number of patients with AKI (serum creatinine >1.5) was 184 (CMC 171 and PIMS 7) giving a prevalence of 21.2% [Figure - 1]. There were more men (141 [76%]) and the mean (SD) age was 41.7 (13.2) years. Most of them were farmers, manual labourers and home-makers. Only 21 patients (11.4%) had any comorbid condition (diabetes, hypertension or both). The mean (SD) duration of stay was 10.3 (8.3) days, with a mean (SD) admission platelet count of 1.34 (0.89)x109/L and mean (sd) admission serum creatinine was 2.78 (1.97) mg/dl. The maximum mean (SD) serum creatinine was 5.62 (3.77) mg/dl and at discharge was 3.59 mg/dl.
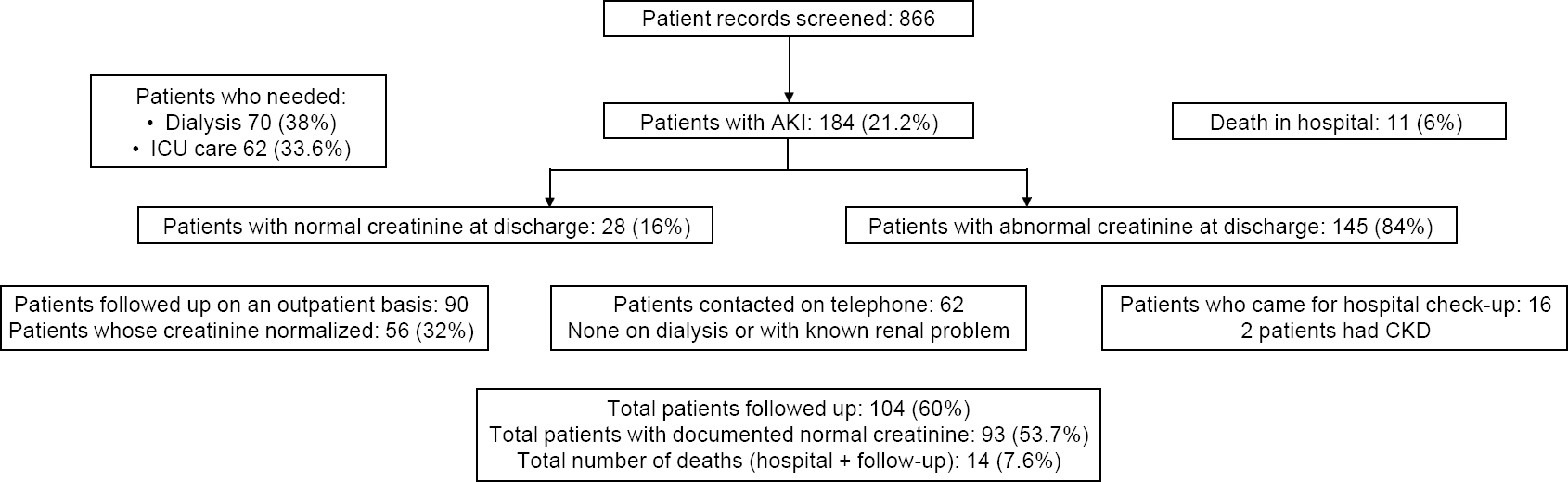 |
| Figure 1: Patients with acute kidney injury (AKI) following snake envenomation and their outcomes |
In our study, the mean (SD) duration of time to attain the maximum creatinine value was 5.4 (5.36) days. The mean (SD) prothrombin time (PT) was 30.7 seconds, and in 19 patients it was >2 minutes. The mean (SD) international normalized ratio was 2.7 (3.0); the mean (SD) activated partial thromboplastin time was 50.6 (48.4) seconds and in 19 patients it was >3 minutes. The mean (SD) maximum creatine kinase was 6765.3 (16 976) IU/L. Urine protein was elevated in 103 (56%) patients assessed by the semi-quantitative dipstick method. Urine white blood cell and red blood cells were elevated by >10 in 33 (18%) patients. Abnormal urine casts were present in 65 (35%) patients, with fine casts in 20, coarse casts in 15 and both in 30 patients.
The snake-envenomed syndromes in our patients were as follows : isolated renal in 21 (11.4%); renal and haematological 5 5 (29.8%); renal and neurological 10 (5.4%); and renal, haematological and neurological 98 (53.2%).
The number of patients admitted to the intensive care unit (ICU) was 62 (33.6%; CMC 55 and PIMS 7). The mean duration of ICU stay was 2.3 (range: 1-18) days with 50 patients requiring only a 1 -day stay. The number of patients put on a ventilator was 44 (23.9%); those on dialysis was 70 (38%; CMC 65 and PIMS 5). The number of patients who died in hospital was 11 (6%; all in CMC). The number of patients with normalized serum creatinine (<1.3 mg/dl) at discharge was 28 (16%) while 145 (84%) had persisting renal dysfunction at discharge. Following up the creatinine values from the records, those who further normalized within the first month of follow-up were 33 (19%). Over the subsequent months, the number of patients with normalized serum creatinine from follow-up of records was 84 (48.5%).
Prospective follow-up
The number of people with phone numbers registered in the hospital records was 115. Of these, 62 (3 6%) people were contacted. We spoke to 21 patients directly and 41 immediate family members. Three patients had died after discharge, 2 of them 3 days after discharge and 1 a month later. All had elevated creatinine at discharge. Of the remaining, none had a known renal disease or were on dialysis. Four of them had new-onset diabetes or hypertension. The mean (SD) follow-up period of these patients was 39 (18) months. Of the 70 patients (3 8%) who needed dialysis during the hospital stay, only 13 patients needed a follow-up for dialysis on an outpatient basis. We were able to contact 8 of these patients and they were doing well, off dialysis without any known kidney problem.
All patients contacted on telephone were requested to come to the hospital for a free health check-up; 16 people came for the review. The mean (SD) follow-up period of these patients was 41 (16) months. Among them, 10 had abnormal serum creatinine at discharge, and during the follow-up period, 9 of them had normalized serum creatinine values. The remaining patient had diabetes with a serum creatinine of 1.4 mg/dl, with active urinary sediments and proteinuria. She had been reviewed 44 months after discharge. Of the 6 patients who had normal serum creatinine values at discharge and follow-up, one patient was a known diabetic and had a creatinine of 2.15 mg/dl with 4+ dipstick proteinuria. He was reviewed 43 months after discharge. Thus, 2 of them developed CKD with associated diabetes.
Binary logistic regression was done to determine the risk factors for death. The variables considered for univariate analysis were age, duration of stay, admission platelet count, maximum creatinine, creatinine at discharge, renal syndrome and dialysis. The variables with p<0.25 were included in the multivariable analyses. Thus, duration of stay, admission platelet and dialysis were included in the multivariable analyses. This revealed that patients who received dialysis had higher odds of death of 5.82 (95% confidence interval [CI] 1.49-22.68) than those who did not. The patients who died had a shorter hospital stay of 5.77 (6.71) days versus 10.75 (8.38) days in those who survived (p=0.004). They also had lower admission platelets with a mean of 0.87 (0.74)x1011/dl in comparison to a mean (SD) of 1.39 (0.89)x1011/dl for patients who survived (p=0.033). Hosmer and Lemeshow statistics (p=0.10) suggested good fit and Nagelkerke R2 was 26%.
Binary logistic regression was also done to compare those who came for a hospital review with those who did not come for a review. No variable was significantly different in these two groups. Hence, we postulated that the outcomes in those who did not come for a review could be comparable to those who did.
Discussion
There are many mechanisms of snake envenoming-induced kidney injury. Hypotension, haemolysis, disseminated intravascular coagulation and direct nephrotoxicity have been described.[18] The renal lesions are usually acute tubular or cortical necrosis. The mechanism of AKI may have an implication in the progression to long-term renal disease, but this has not been established.
In our study, AKI was seen in 21.2% of patients. Other studies show a wide range of incidence of AKI with snake envenoming [Table - 1]. This can be due to the regional variation in snake species, and also local practices that may delay patients from reaching healthcare facilities. In our study, over 70% of patients reached the hospital within 1 day and did not resort to prolonged treatment from native healers. The need for dialysis in patients with AKI is high and similar to the trends in the rest of India. The hospital mortality in our study was lower than that of other studies. This is possibly due to the earlier presentation, appropriate antivenom administration, early dialysis and good quality of ICU care. The antivenom that is nationally available and used in these centres are developed in Tamil Nadu, from regional snake species, and hence it may be more effective in its neutralizing properties. This hypothesis, however, needs further evaluation.

Data on long-term renal outcomes in snake envenomation- induced AKI are scarce. Waikhom et al. prospectively followed up 60 patients who developed AKI requiring dialysis following a snake bite for a mean of 45 months; 41% had a persistent renal dysfunction with 3 patients progressing to CKD Stage V.[11] In a study from Sri Lanka, Herath et al. followed up 54 patients with AKI following snake bite for 1 year.[12] Of these patients, 37% developed CKD and 5 progressed to end-stage renal disease. We could prospectively follow-up 61% of patients (a composite of creatinine from hospital records, telephonic interview or hospital visit) and found that only 2 had documented raised creatinine and none were on dialysis. Even the 2 with CKD had diabetes and hence we cannot directly attribute the renal injury to the episode of AKI. Although those who were telephonically contacted did not report any known renal disease, it is difficult to be sure that they did not have renal dysfunction. No patients were on dialysis, and it is possible that the outcome may not be as adverse in this region as reported by previous studies. The commonly observed syndrome in our patients was renal, neurological and haematological (53%) followed by renal and haematological (30%). It is likely that the biting species causing AKI was Russell’s viper. The hump-nosed viper is not a biting species in our region and is found mostly in western parts of India and in Sri Lanka. Although a similar syndrome can be caused by the Hump- nosed pit viper, it is not a biting species in this area.
Limitations
We did not have the premorbid creatinine values for most patients, and hence could not rule out prior CKD. Only a proportion of patients were reachable by telephone since most of the contact numbers were either out of use or transferred. A telephonic interview can provide only partial information on a patient’s clinical problems, and laboratory evaluation is essential in following up patients with renal involvement. The data are from two centres and the patient profile in these centres reflects a tertiary care experience, which cannot be generalized.
Conclusion
AKI with snake envenomation is a common problem. This affliction impacts mostly middle-aged men from a lower socioeconomic class. If treated appropriately with anti-snake venom, intensive monitoring and prompt dialysis, a majority of patients will recover. We also observed that most patients do not progress to symptomatic renal morbidity and are not on dialysis. Thus, further prospective validation is warranted and centres should promote appropriate intensive therapy that can be life-saving.
Conflicts of interest. None declared
Acknowledgement
We thank Dr David A.Warrell for his input and suggestions in preparing the manuscript.
| 1. | Kasturiratne A, Wickremasinghe AR, de Silva N, Gunawardena NK, Pathmeswaran A, Premaratna R, et al. The global burden of snakebite: A literature analysis and modelling based on regional estimates of envenoming and deaths. PLoS Med 2008;5:e218. [Google Scholar] |
| 2. | Mohapatra B, Warrell DA, Suraweera W, Bhatia P, Dhingra N, Jotkar RM, et al. Snakebite mortality in India: A nationally representative mortality survey. PLoS Negl Trop Dis 2011;5:e1018. [Google Scholar] |
| 3. | Narvencar K. Correlation between timing of ASV administration and complications in snake bites. J Assoc Physicians India 2006;54:717-19. [Google Scholar] |
| 4. | Harshavardhan L, Lokesh AJ, Tejeshwari HL, Halesha BR, Metri SS. A study on the acute kidney injury in snake bite victims in a tertiary care centre. J Clin Diagn Res 2013;7:853-6. [Google Scholar] |
| 5. | Bhalla G, Mhaskar D, Agarwal A. A study of clinical profile of snake bite at a tertiary care centre. Toxicol Int 2014;21:203-8. [Google Scholar] |
| 6. | Athappan G, Balaji MV, Navaneethan U, Thirumalikolundusubramanian P. Acute renal failure in snake envenomation: A large prospective study. Saudi J Kidney Dis Transpl 2008;19:404-10. [Google Scholar] |
| 7. | Suchithra N, Pappachan JM, Sujathan P. Snakebite envenoming in Kerala, South India: Clinical profile and factors involved in adverse outcomes. Emerg Med J 2008;25:200-4. [Google Scholar] |
| 8. | Ariaratnam CA, Sheriff MH, Arambepola C, Theakston RD, Warrell DA. Syndromic approach to treatment of snake bite in Sri Lanka based on results of a prospective national hospital-based survey of patients envenomed by identified snakes. Am J Trop Med Hyg 2009;81:725-31. [Google Scholar] |
| 9. | Kularatne SA, Silva A, Weerakoon K, Maduwage K, Walathara C, Paranagama R, et al. Revisiting Russell’s viper (Daboia russelii) bite in Sri Lanka: Is abdominal pain an early feature of systemic envenoming? PLoS One 2014;9:e90198. [Google Scholar] |
| 10. | Warrell DA. Guidelines for the management of snake-bites. Geneva:WHO; 2010. [Google Scholar] |
| 11. | Waikhom R, Sircar D, Patil K, Bennikal M, Gupta SD, Pandey R, et al. Long-term renal outcome of snake bite and acute kidney injury : A single-center experience. Ren Fail 2012;34:271^. [Google Scholar] |
| 12. | Herath HM, Wazil AW, Abeysekara DT, Jeewani ND, Weerakoon KG, Ratnatunga NV, et al. Chronic kidney disease in snake envenomed patients with acute kidney injury in Sri Lanka: A descriptive study. Postgrad Med J 2012;88:138^42. [Google Scholar] |
| 13. | Warrell DA. Geographical and intraspecies variation in the clinical manifestations of envenoming by snakes. In: Thorpe RS, Wuster W, Malhotra A (eds). Venomous snakes: Ecology, evolution and snakebite. Oxford:Clarendon Press; 1997:189-203. Available at www.tib.eu/en/search/id/BLCP%3ACN016816123/Geographical-and- intraspecies-variation-in-the/?tx_tibsearch_search%5Bsearchspace%5D=tn# documentinfo (accessed on 14 Jun 2017). [Google Scholar] |
| 14. | Chippaux JP, Williams V, White J. Snake venom variability: Methods of study, results and interpretation. Toxicon 1991;29:1279-303. [Google Scholar] |
| 15. | Chauhan V, Thakur S. The North-South divide in snake bite envenomation in India. J Emerg Trauma Shock 2016;9:151^4. [Google Scholar] |
| 16. | Acute Kidney Injury Work Group. Kidney disease: Improving global outcomes (KDIGO) - Clinical practice guideline for acute kidney injury. Kidney Int 2012;2: 1-138. [Google Scholar] |
| 17. | KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. National Guideline Clearinghouse. Available at www.guideline.gov/summaries/summary/46510/kdigo-2012-clinical-practice- guideline-for-the-evaluation-and-management-of-chronic-kidney-disease (accessed on 14 Jun 2017). [Google Scholar] |
| 18. | Kohli HS, Sakhuja V. Snake bites and acute renal failure. Saudi J Kidney Dis Transpl 2003;14:165-76. [Google Scholar] |
| 19. | Ali G, Kak M, Kumar M, Bali SK, Tak SI, Hassan G, et al. Acute renal failure following Echis carinatus (saw-scaled viper) envenomation. Indian J Nephrol 2004;14:177-81. [Google Scholar] |
| 20. | Dharod MV, Patil TB, Deshpande AS, Gulhane RV, Patil MB, Bansod YV, et al Clinical predictors of acute kidney injury following snake bite envenomation. N Am J Med Sci 2013;5:594-9. [Google Scholar] |
| 21. | Mukhopadhyay P, Mishra R, Mukherjee D, Mishra R, Kar M. Snakebite mediated acute kidney injury, prognostic predictors, oxidative and carbonyl stress: A prospective study. Indian J Nephrol 2016;26:427-33. [Google Scholar] |
Fulltext Views
2,128
PDF downloads
1,089




